Diagnosis and Management of Ecthyma Gangrenosum in Chronic Renal Failure Patient
Article information
Ecthyma gangrenosum (EG) is an aggressive cutaneous disease caused by local or systemic Pseudomonas aeruginosa infection characterized by cutaneous manifestations ranging from maculopapular lesions and hemorrhagic bullae to necrotic tissue ulceration with surrounding erythema [1,2,3,4,5]. The differential diagnosis includes other causes of necrotic wounds such as calciphylaxis, septic emboli, cutaneous anthrax, cutaneous aspergillosis, and pyoderma gangrenosum. Individuals with EG are most often actively immunocompromised [3,4,5]. A high degree of clinical suspicion is required, but a tissue sample is ultimately necessary for definitive diagnosis. Current treatment recommendations include wide local excision as soon as a diagnosis is made concurrent with antibiotic therapy [3,4]. Reconstructive options vary and are largely dependent on the resultant defect's size, depth, and location [3]. While this disease has been described in various populations, the surgeon's role in EG is not well elucidated (Supplemental Table S1), notably in the adult population [1,2,3,4,5]. We present a case report emphasizing the surgeon's role in the diagnosis and management of EG in an adult with chronic renal failure.
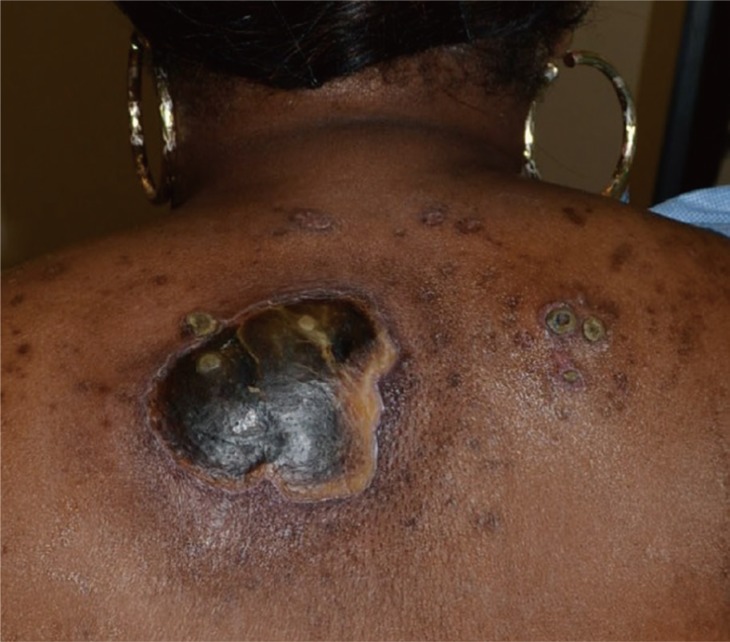
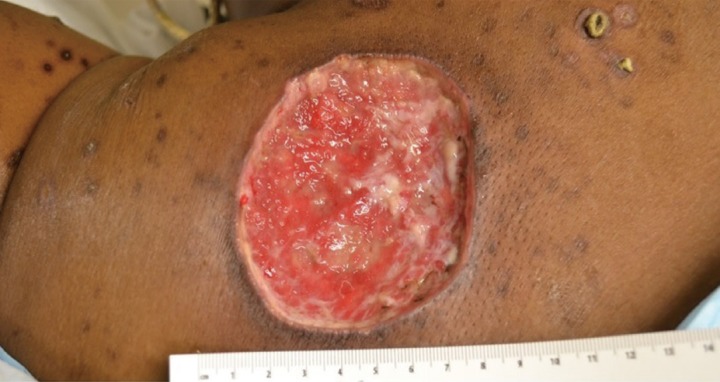
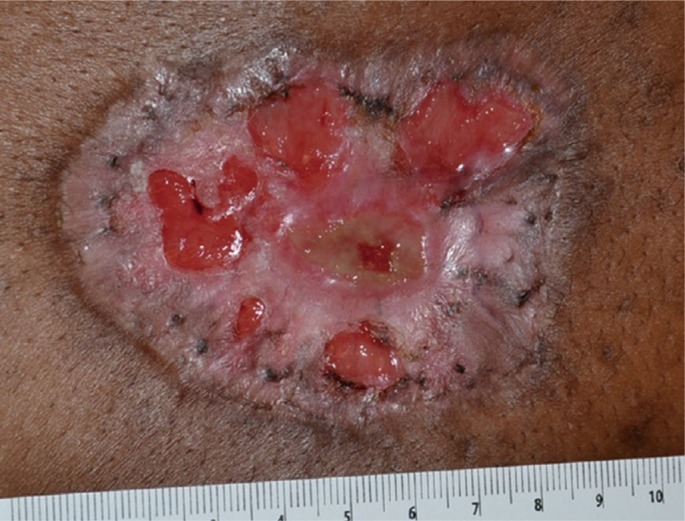
A 46-year-old black woman with end-stage renal disease secondary to insulin dependent diabetes on hemodialysis for seven years, hypertension, peripheral vascular disease necessitating an above-knee amputation, and previous stroke presented to the Emergency Department with a 3 centimeter painless papular lesion on her midline upper back for approximately one month. She receives hemodialysis through a left-sided internal jugular hemodialysis catheter; a right-sided catheter had been removed for infection. Approximately six weeks after, she returned to the Emergency Department with progression of the same lesion despite the addition of an IV antibiotic regimen. A painless 8 centimeter ulcer with fibrinous borders and a black eschar base without drainage or surrounding erythema was found (Fig. 1). She was referred to the Wound Center's plastic surgeon and found to have an 8 cm ulcer with a moist eschar which had begun to drain yellow fluid. Surgical biopsy and debridement was undertaken approximately one week later (Fig. 2). Her white blood cell count was 11,500/µL, calcium was 8.9 mg/dL and creatinine was 3.1 mg/dL. Other laboratory findings were non-contributory. A 7×9 centimeter lesion was excised with 5 mm margins. She was begun on Clindamycin and Ciprofloxacin with wet to dry dressing changes with Dakin's solution for her resultant wound. Dakin's solution was chosen for its antibacterial properties. One month postoperatively, the patient's wound dressing regimen was changed from wet to dry dressing changes with Dakin's solution to silver-impregnated alginate dressing, to allow for improved exudate absorption (Fig. 3).
Postoperatively, her wound cultures grew Pseudomonas aeruginosa and Staphylococcus epidermidis. Her pathology report indicated "a broadly ulcerated zone of necrotic dermis and subcutis with a dense predominately neutrophilic inflammatory infiltrate, abundant bacteria and abscess formation... acute suppurative inflammation extends to the edge of the debridement (which) suggest(s) EG." Postoperatively, she has not experienced disease progression or recurrence and is in her usual state of health. She has elected to continue with dressing changes despite being offered negative pressure wound therapy and/or split-thickness skin grafting for wound coverage.
The presented case is unique for a variety of reasons. As previously discussed, EG is frequently a progressive and possibly fatal disease with characteristic cutaneous manifestations and affected populations [1,2,3,4,5]. Despite having chronic renal failure on hemodialysis, the presented patient was not neutropenic and did not exhibit other manifestations of an immunocompromised state as associated with EG in previous reports [3,4,5]. Other potential etiologies include calciphylaxis, localized necrotic infection, given the surrounding excoriations, and superinfected pressure ulceration. Of these, calciphylaxis was most strongly considered as an etiology prior to pathology results, given her renal status and wound appearance as well as evidence of extensive vascular calcification on imaging studies. While the patient did not have a bone density study on record, evidence of diffuse demineralization is seen on imaging.
The etiology of EG is well-established as Pseudomonas aeruginosa infection often secondary to immunosuppression. Given a lack of systemic infectious symptoms, a local inoculation was the most likely harbinger of this patient's presentation; although seeding from hemodialysis catheter infection cannot be ruled out given the patient's history. However, the presented case suggests the possibility of a synergistic interaction between the primary infectious and immunosuppressive etiology of the disease and a patient's underlying vascular status. The patient's diffuse vascular compromise, as evidenced by her history of an above-knee amputation and stroke, may have placed her at higher risk for presenting with cutaneous manifestations after Pseudomonas aeruginosa inoculation. In this case, the interaction between the infectious etiology of EG and the patient's vascular status cannot be fully ascertained. However, this is the first report to suggest this link.
The surgeon's role in providing definitive treatment for EG is defined in the literature. Yet their role in the diagnosis of EG, especially in equivocal, idiopathic cases, has not been well-documented as evidenced by Supplemental Table S1. The surgeon must have a low threshold to perform excisional biopsy for culture and pathology in all cases of undiagnosed wounds; however, this case demonstrates the unique prudence of this principle in lesions suspicious for or suggestive of EG. As evidenced by this case report, there is a high risk of misdiagnosis in cases of EG.
Reconstructive options for secondary defects in cases of EG have not been as well described in the adult population as in the pediatric population [2,3]. There is no "gold standard" for reconstruction of these defects given the variability in their presentation. Defect location is a primary determinant of the option chosen as is defect size, depth, and patient preference. As a cutaneous disease that does not typically affect underlying structures, healing by secondary intention, local flap coverage, and skin grafting are all valid options after debridement and biopsy in EG. This patient was offered a wound vacuum system, skin graft, and healing by secondary intention for treatment of her wound and elected healing by secondary intention, which has thus far yielded acceptable results.
In conclusion, we present a case demonstrating the important role played by the surgeon in the definitive diagnosis and treatment of EG in an adult patient with chronic renal failure.
Notes
This article contains supplemental Table S1.
No potential conflict of interest relevant to this article was reported.
References
Supplementary Material
Supplemental Table S1
Literature review-ecthyma gangrenosum and the surgeon's role
Supplemental data can be found at: http://e-aps.org/src/sm/aps-41-299-s001.pdf