Management of complex surgical wounds of the back: identifying an evidence-based approach
Article information
Abstract
Background
Postoperative dehiscence and surgical site infection after spinal surgery can carry serious morbidity. Multidisciplinary involvement of plastic surgery is essential to minimizing morbidity and achieving definitive closure. However, a standardized approach is lacking. The aim of this study was to identify effective reconstructive interventions for the basis of an evidence-based management protocol.
Methods
A retrospective review was performed at a single tertiary institution for 45 patients who required 53 reconstruction procedures with plastic surgery for wounds secondary to spinal surgery from 2010 to 2019. Statistical analysis was performed for demographics, comorbidities, and treatment methods. Primary outcomes were postoperative complications, including dehiscence, seroma, and infection. The secondary outcome was time to healing.
Results
The overall complication rate was 32%, with dehiscence occurring in 17%, seroma in 15% and infection in 11% of cases. Median follow-up was 10 months (interquartile range, 4–23). Use of antibiotic beads did not affect rate of infection occurrence after wound closure (P=0.146). Use of incisional negative pressure wound therapy (iNPWT) was significant for reduced time to healing (P=0.001). Patients treated without iNPWT healed at median of 67.5 days while the patients who received iNPWT healed in 33 days. Demographics and comorbidities between these two groups were similar.
Conclusions
This data provides groundwork for an evidence-based approach to soft tissue reconstruction and management of dehiscence after spinal surgery. Timely involvement of plastic surgery in high-risk patients and utilization of evidence-based interventions such as iNPWT are essential for improving outcomes in this population.
INTRODUCTION
Postoperative dehiscence and surgical site infection (SSI) are significant complications of spinal surgery, leading to increased morbidity and mortality [1]. In this patient population, the incidence of isolated wound dehiscence is 0.3%, while the rate of wound dehiscence with infection is 2.5% based on National Surgery Quality Improvement Program data [2]. According to the National Nosocomial Infections Surveillance System, SSI occurs in 1% of discectomies and laminectomies, 2%–5% of fusions, and up to 8.5% of fusions when an implant is placed [3].
Prevention of SSI not only improves clinical outcomes but has significant savings benefits. The average cumulative cost of readmission and treatment of SSI after spinal surgery is $16,242, in addition to the total cost of care [4]. More specifically, patients with SSI following surgery for spinal neoplasms incur inpatient hospital costs 60% higher compared to those without SSI [5]. Occurrence of SSI was found to nearly double length of stay and increase the likelihood of readmission by more than six times compared to general surgery patients experiencing no infectious complications [6].
Postoperative dehiscence in spinal surgery patients also greatly increases the rate of readmission (odds ratio [OR], 19.6; P < 0.01) and reoperation (OR, 23.6; P < 0.01) [7]. Known risk factors for dehiscence after spinal surgery include diabetes, obesity, smoking, history of radiotherapy, and hypertension [3,8-10]. Operative factors such as a posterior approach, involvement of seven or more intervertebral levels, instrumentation, prolonged operative time, and revision spinal surgery also predispose to healing complications [3,8-12]. Common approaches to decrease dehiscence include antibiotics, surgical debridement, muscle flaps, and negative pressure wound therapy or a combination thereof [13]. Effective multidisciplinary management with early involvement of plastic surgery is essential to minimize morbidity and mortality in the event of dehiscence and achieve definitive closure in this comorbid population. However, there is currently no standard protocol for the management of wound dehiscence after spinal surgery.
We present a retrospective review of our institutional experience with management of complex wounds following spinal surgery over a 10-year period. The aim of this study was to identify effective interventions to provide the basis for an evidencebased management protocol.
METHODS
Retrospective review
After receiving IRB approval (IRB No. 2018-173), a single center retrospective review was performed of all consecutive patients who required plastic surgery intervention after spine surgery. Written informed consents were obtained. Soft tissue reconstruction and closure was performed by one of three senior authors (CEA, KKE, and SRS).
Data collected included patient demographics, comorbidities, and perioperative labs. Details of prior spinal surgery (type, operative time, bone graft use, spinal hardware use) were also identified. Operative variables for plastic surgery intervention were also documented including number of debridements prior to closure and reconstructive approach.
Outcomes of interest
The primary outcome of interest was the rate of postoperative complications following soft tissue reconstruction, including hematoma, dehiscence, and infection. Infection was defined as clinical signs of infection confirmed by positive wound cultures. Complications directly associated with the preceding spine procedure (e.g., cerebrospinal fluid leak) and medical complications were excluded from analysis. Both the overall incidence of complications and rates of individual complications (dehiscence, seroma, and infection) were analyzed. The secondary outcome of interest was time to healing.
Statistical analysis
Descriptive statistics were used to describe study subjects. Continuous variables were described by means with standard deviation or medians with interquartile ranges (IQRs) as appropriate based on the Shapiro-Wilk test of normality. Categorical variables were described by frequencies and percentages.
Logistic regression was performed to examine the relationship between binary outcomes (overall complications, dehiscence, seroma, infection) and explanatory variables. Multivariate logistic regression was not performed due to inadequate power as determined by the 10 events per variable rule [14].
Patients who failed to heal and required reoperation after postoperative day 90 were censored at the date of reoperation and the second reconstruction was included in analysis as a unique procedure. Cox proportional hazards regression was utilized to test the association between explanatory variables and the endpoint of healing, specifically to assess their effects on healing over time while accounting for confounding factors. All predictors with P < 0.200 on bivariate analysis were included in the multivariate regression analysis using a backwards model. Kaplan-Meier analysis was used to graphically display time to healing. The Schoenfeld residuals method was utilized to check the proportional hazards assumption.
Statistical analysis was performed using STATA version16 (StataCorp, College Station, TX, USA) with significance defined as P < 0.05.
RESULTS
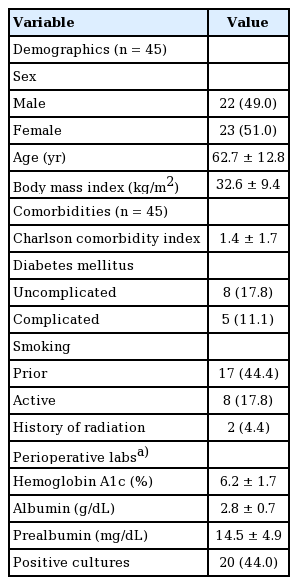
During the study period, a total of 45 patients underwent 53 soft tissue reconstruction procedures with plastic surgery for complex wounds secondary to spinal surgery. On average, the population was obese (mean body mass index, 32.6 kg/m2) and with a mean Charlson comorbidity index of 1.4 (Table 1) [15]. The Charlson comorbidity index is a comorbidity-based score ranging between 0 and 24 used to predict mortality. A score of 1.4 correlates to a 10-year survival rate between 90% and 96%. Common comorbidities were diabetes and smoking. Perioperative labs are presented in Table 1; however, they were documented inconsistently. Day of reconstructive qualitative cultures were positive in 20 cases.
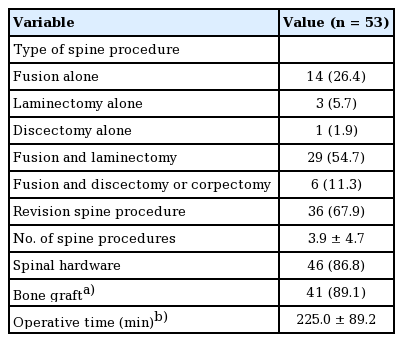
The most common preceding spine procedure was spinal fusion with laminectomy in 29 cases. Other procedures included fusion alone, laminectomy, discectomy, and a combination of fusion and another procedure besides or in addition to laminectomy (Table 2). The majority of cases (86.8%) had spinal hardware at time of reconstruction; in four cases the patient had a history of hardware but underwent complete removal of hardware prior to reconstruction. The median operative time of the preceding spine procedure was 225 minutes.
Reconstruction outcomes
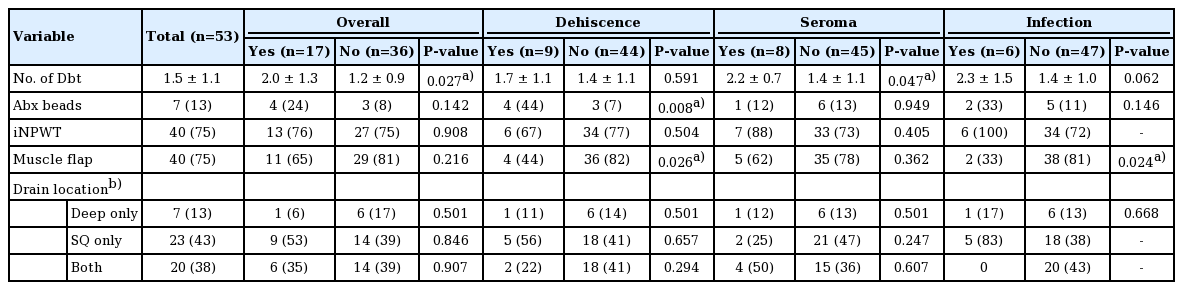
Approach to soft tissue reconstruction and closure is described in Table 3. The average number of debridements prior to reconstruction was 1.5. Antibiotic beads were placed or retained at time of reconstruction in seven patients (13%). A local muscle flap was utilized in 40 of cases (75%). Specifically, paraspinal advancement flaps were used in 34 (64%) and trapezius flaps in 10 cases (19%); the latissimus dorsi was used in one case (2%). Incisional negative pressure wound therapy (iNPWT) was utilized in 40 cases (75%).
The overall complication rate was 32% of cases. On individual analysis, dehiscence occurred in nine (17%), seroma in eight (15%) and infection in six (11%) cases. On bivariate logistic regression, only number of debridements was found to have a significant relationship with overall complications (Table 3). Each additional debridement increased the odds of a complication by 2.0 times (95% confidence interval [CI], 1.1–3.7; P = 0.027). Number of debridements also exhibited a significant relationship with incidence of seroma (OR, 2.1; 95% CI, 1.0–4.5; P = 0.047). There was no significant relationship between drain location and seroma (Table 3). Both use of antibiotic beads and muscle flaps were significant for dehiscence. Antibiotic beads increased the odds of dehiscence by 10.9 times (95% CI, 1.9–63; P = 0.008). Comparatively, use of muscle flap was found to decrease the odds of dehiscence by a factor of 5.6 (95% CI, 1.2–26; P = 0.026). Use of a muscle flap was also significant for decreased odds of infection (OR, 8.4; 95% CI, 1.3–53; P = 0.024); however, use of antibiotic beads did not demonstrate a significant relationship with infection (P = 0.146).
Bivariate logistic regression was also performed with patient demographics, comorbidities, and characteristics of spine procedures as explanatory variables to assess for potential confounders. Complicated diabetes was statistically significant for dehiscence and infection (P = 0.046 and P = 0.026, respectively). Hemoglobin A1c was also significant for infection (P = 0.020). No other variables reached significance (see Supplemental Table 1, which provides p-values for all possible confounders considered). Multivariate logistic regression was not performed due to inadequate power; thus, Fisher exact test was used to assess for an association between use of antibiotic beads or use of muscle flap and diabetes as these were the reconstructive variables found to be significant for dehiscence and infection. There was no statistically significant association between use of antibiotic beads and diabetes (P = 0.549) or use of muscle flap and diabetes (P = 0.097). Similarly, Wilcoxon rank-sum test was used to compare A1c between muscle flap and non-muscle flap groups and was not significantly different (P = 0.748).
Time to healing
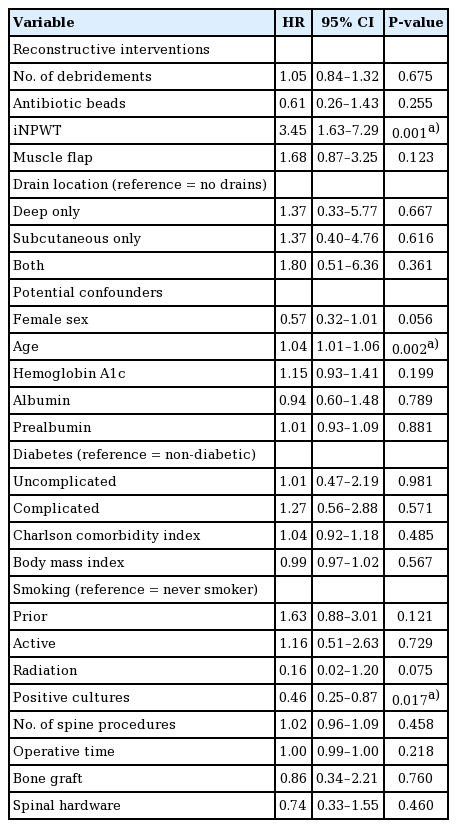
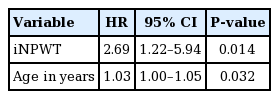
Healing was observed in 51 cases with a median time to healing of 36 days (IQR, 27–48). In two cases, healing was not achieved and the patients underwent a second attempt at reconstruction after POD90; the three recurrences occurred at a mean of 1.3 months (standard deviation, 1.0) after initial healing. Two of the recurrences occurred in the same patient; healing was achieved after complete removal of hardware. In the second patient experiencing recurrence, healing was achieved with use of NPWT. Median follow-up duration was 10 months (IQR, 4–23 months). Bivariate Cox hazards regression was performed (Table 4). The only reconstructive intervention with significant effect was iNPWT (hazard ratio [HR], 3.4; 95% CI, 1.6–7.3; P = 0.001); at any given point in time, 3.4. The median time to healing in the iNPWT versus non-iNPWT groups was 33 days (IQR, 26– 42) and 67.5 days (IQR, 44.5–276), respectively (Fig. 1). Potential confounders exhibiting significance on bivariate analysis were age (P = 0.002) and positive day of reconstruction cultures (P = 0.017).
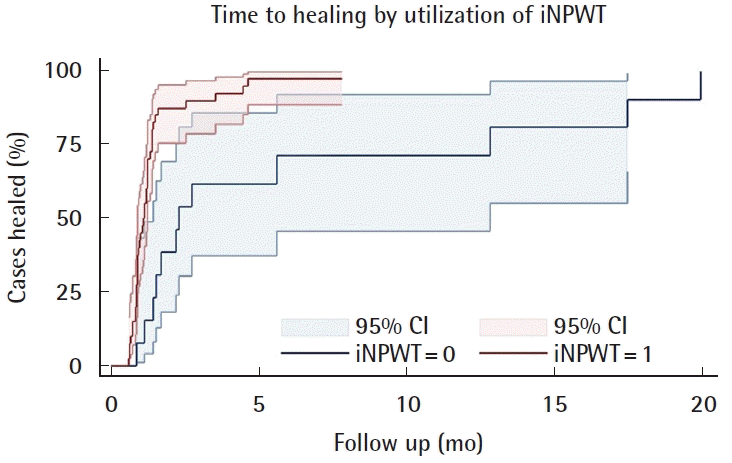
Healing in iNPWT versus non-iNPWT groups. A Kaplan-Meier failure curve for time to healing in patients treated with incisional negative pressure wound therapy (iNPWT=1) versus without (iNPWT=0). The iNPWT group had a 73% probability of achieving healing earlier than the non-iNPWT group. iNPWT, incisional negative pressure wound therapy; CI, confidence interval.
The final results of multivariate Cox hazards regression model are presented in Table 5. Both iNPWT (HR, 2.7) and age (HR, 1.03) maintained independently significant relationships with time to healing. Positive day of reconstruction cultures was dropped from the model due to lack of significance.
DISCUSSION
This retrospective series of patients requiring plastic surgery closure for dehiscence after spinal surgery identifies a population that is predominantly diabetic and obese. A large portion of the patients also presented with current or prior tobacco use and a history of revision spine surgery. The mean operative time for the patients was prolonged ( > 3 hours). Use of iNPWT was the only intervention found to have a significant relationship with decreased time to healing.
Identification of these risk factors is consistent with prior literature. Two meta-analyses identified diabetes, obesity, and prolonged operative time as significant risk factors for SSI after spinal surgery [3,8]. In a pooled analysis of patients from four studies, smoking and diabetes were also identified as risk factors for development of complex back wounds requiring muscle flaps for coverage [9]. In a group of 2,391 patients who developed postoperative spinal wound infection, 37% of the patients had undergone revision spinal surgery [12]. Using these factors, high-risk patients who would benefit from multidisciplinary evaluation can be identified and considered for prophylactic soft-tissue intervention. However, a standardized approach to management of this population is lacking.
We found that use of antibiotic beads has no effect on the rate of infectious complications after wound closure. A prior study by Horii et al. [16] also found no difference in the incidence of developing SSI after spinal surgery between patients treated with or without antibiotic powder. In some cases, we observed persistent infection when utilizing antibiotic beads. In one of the patients who experienced recurring infection in our series, antibiotic beads had been placed multiple times and infection continued to recur, precluding successful closure. An in vitro study by Howlin et al. [17] found that antibiotic beads had limited efficacy in treating established biofilms in periprosthetic infections. Furthermore, their data from scanning electron microscopy demonstrated potential for antibiotic beads to act as a nidus for biofilms once antibiotic concentrations fell below minimum bactericidal concentration. The potential of antibiotic beads to act as a nidus for infection is especially concerning given the lack of demonstrated significance for improving the incidence of infectious complications. In the cases of recurrent infection, we also observed instances of sinus tract infection while using antibiotic beads. Although this observation was anecdotal, our analysis found that antibiotic beads were significantly associated with increased rates of wound dehiscence, which warrants further investigation of the deleterious impact antibiotic beads may have on primary wound closure.
Each debridement a patient received increased the odds of a complication by 2.0 times, which likely is the result of increased surgical trauma. However, the number of debridements may also be indicative of a more infectious wound base or poor tissue quality preventing proper wound healing. With each serial debridement, more tissue is excised, increasing the diameter and the depth of the wound. Increased dead space is an independent risk factor for seroma formation [18], which is further demonstrated by the significant correlation between number of debridements and risk of seroma development in our study. Surgical drains are routinely installed to prevent seroma formation, whether subcutaneously or deep to the fascial layer. Results from our analysis found no difference in seroma rates with the use of drains, regardless of location. Seromas can occur after drain removal, especially if the drain is removed too early [19]. While most seromas can be treated conservatively, more refractory seromas may require more intensive management, such as sclerotherapy [20]. To prevent late-occurring seromas, our institution has begun to utilize sclerosing agents within surgical drains, to ensure dead space elimination, prior to drain removal in patients with history of recurrent seromas.
Immediate soft tissue reconstruction of complex wounds secondary to spinal surgery with muscle flaps is associated with high rates of successful wound closure while minimizing wound complications [21]. The results of our study further support this claim, with muscle flaps significantly decreasing rates of dehiscence and infectious complications postoperatively. The use of well-vascularized muscle flaps provides ample coverage of any exposed hardware as well as increases delivery of systemic antibiotics. Furthermore, the multiple layers of tissue within the flap minimize hardware exposure in the case of minor wound breakdown [21].
We found a significantly reduced time to healing in patients treated with iNPWT. Patients who were treated with iNPWT had a 73% probability of healing prior to those without iNPWT [22]. Similarly, a randomized control trial of patients who developed wounds after surgery for spinal fractures found that the use of iNPWT was shown to significantly reduce mean seroma volume, wound care time, number of dressings required, and duration of wound secretion [23]. In a study of patients who underwent long-segment thoracolumbar spinal fusions, the group of patients who received prophylactic iNPWT had a significant reduction of both wound dehiscence and postoperative SSIs compared to the patients treated without prophylactic iNPWT [24]. Increasing age was associated with improved odds of earlier healing in our study; a 10-year increase in age results in a 57% probability of healing earlier (coefficient = 0.028) while holding all other variables constant [22]. Our series included one patient 25 years of age with healing occurring after 24 months, far younger than our median of 62 years with healing time far longer than our median of 36 days; inclusion of this patient may have skewed analysis, warranting further investigation.
We advocate for the following evidence-based treatment algorithm (Fig. 2). After a thorough physical exam and assessment of medical comorbidities, the initial evaluation should focus on signs of infection. If there is suspicion for infection, wound cultures should be taken immediately prior to initiation of antibiotic therapy. If positive, then debridement should ensue and an infectious disease specialist should be consulted in order to select proper antibiotic therapy. Cultures should again be taken after debridement to confirm an absence of infection. This cycle of debridement and cultures should be repeated until the wound cultures are negative. Based on our findings, we do not advocate for the use of antibiotic beads. Residual infection should be addressed with further debridement and/or hardware removal when appropriate.
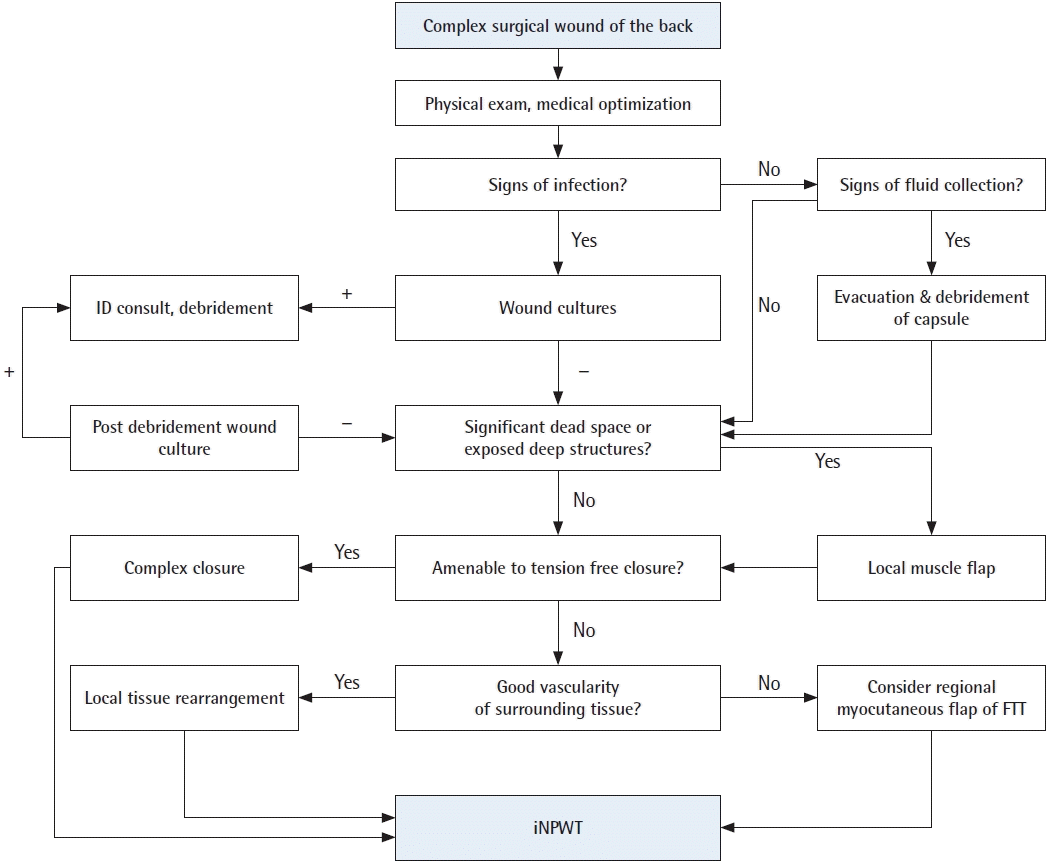
Evidence-based management algorithm for complex back wounds. An evidence-based management algorithm for complex surgical wounds of the back to guide infection control, treatment of fluid collections, and dead space elimination. Following these steps, all wounds should then be managed with incisional negative pressure wound therapy (iNPWT) for a minimum of 7 days. FTT, free tissue transfer.
After addressing infection and any fluid collection, the defect should be examined to plan for soft tissue reconstruction. Defects with significant dead space or exposed deeper structures, including hardware, necessitate muscle flap coverage. We found no significant difference in outcomes between local flap options including paraspinal and trapezius flaps. Next, the quality and laxity of the soft tissue must be considered. Given the deleterious effects of additional healing complications, it is paramount to ensure a tension-free closure.
We have found that local tissue rearrangement is sufficient to achieve definitive closure in almost all cases when primary closure is not possible. There were no patients that required free tissue transfer in this study. A history of radiation therapy to the surgical site requires wide resection of irradiated tissue and often results in a large skin and muscle defect that may not be amenable to local tissue rearrangement. Free tissue transfer may be considered; however, lack of recipient vessels and need for prone positioning of the patient limit the widespread applicability of this modality. Other options include regional myocutaneous flaps outside the zone of injury, depending on the location of the wound [25,26]. All wounds should then be managed with iNPWT (Fig. 3) for a minimum of 7 days postoperatively regardless of defect size or approach to reconstruction as this was the only intervention found to decrease time to healing in our series.
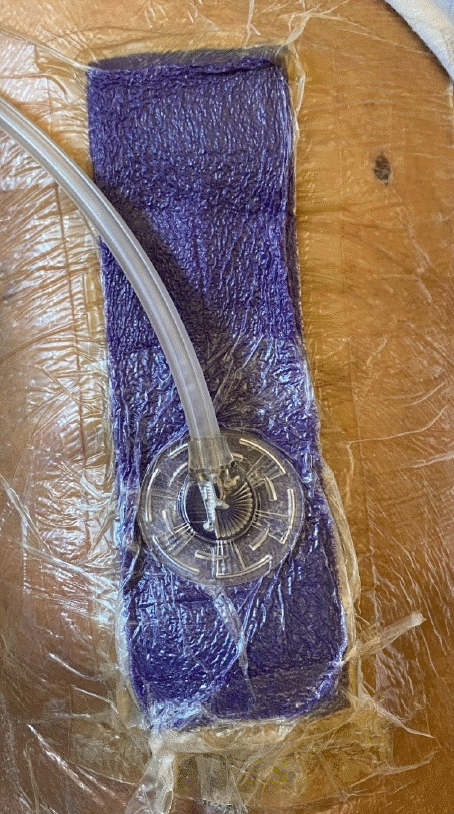
Incisional negative pressure wound therapy (iNPWT) dressing. Example of iNPWT dressing utilized to create physical barrier from contamination, bring incision edges together, and remove excess fluid and infectious material.
This study establishes groundwork for the first evidence-based algorithm for soft tissue reconstruction in the spinal surgery population. However, the retrospective nature of the study did limit analysis. Preoperative factors such as the time to presentation and number of prior spinal operations were often poorly documented. Furthermore, the sample size for certain interventions, such as antibiotic bead placement, was small which also limits the strength of our analysis. Further investigation is warranted to further validate our treatment algorithm for soft tissue reconstruction in this population and would benefit from larger, multicenter studies.
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Ethical approval
The study was approved by the Institutional Review Board of MedStar Georgetown University Hospital (IRB No. 2018-173) and performed in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained.
Author contribution
Conceptualization: EG Zolper, MA Saleem, KG Kim, MD Mishu, SR Sher, CE Attinger, KL Fan, KK Evans. Data curation: EG Zolper, MA Saleem, MD Mishu. Formal analysis: EG Zolper. Methodology: EG Zolper, SR Sher, CE Attinger, KL Fan, KK Evans. Project administration: EG Zolper, KG Kim, SR Sher, CE Attinger, KL Fan, KK Evans. Writing - original draft: EG Zolper, MA Saleem, KG Kim, MD Mishu, SR Sher. Writing - review & editing: EG Zolper, MA Saleem, KG Kim, MD Mishu, SR Sher, CE Attinger, KL Fan, KK Evans.
Supplementary material
Supplemental Table 1. Complication rate by possible confounding variables. Supplemental data can be found at: https://doi.org/10.5999/aps.2020.02061