Two-Blade Guillotine Technique for Nipple Graft Harvest
Article information
Abstract
The nipple-sharing technique for nipple reconstruction offers excellent tissue matching. The method used for nipple graft harvesting determines the quality of the graft and hence, the success of nipple sharing. Here, we described a guillotine technique wherein the nipple is first transfixed with 2 straight needles to stabilise it. Two No. 11 blades are then inserted in the center and simultaneously swept outwards to amputate the distal portion of the nipple. This technique provides good control, resulting in a very evenly cut base. The recipient bed is deepithelialized thinly, and the nipple graft is inset with interrupted 8-0 nylon sutures under magnification. Being a composite graft, it is protected with splint dressings for 6 weeks, and the dressing is regularly changed by the surgeon. The height of the nipple grafts ranges from 4 to 8 mm. This technique was performed in 9 patients with an average follow-up of 2.9 years (range, 1–4.5 years). Apposition between the nipple graft and its bed is crucial for the success of this technique. When correctly applied, we observed rapid revascularization of the graft.
INTRODUCTION
The nipple-sharing technique for nipple reconstruction offers excellent tissue matching [1-4]. The indication of this method is the sufficiency of the contralateral nipple, and importantly, the patient must accept it since not many women are prepared to undergo scarring on their normal side [5-7]. Nipple grafts are susceptible to failure because of ischemia related to the volume of the grafted tissue [8,9]. Therefore, most surgeons recommend taking no more than 5 mm of the nipple when using the nipple-sharing method. In this paper, we describe the maximization of nipple graft take with a guillotine harvest method.
IDEA
Technique
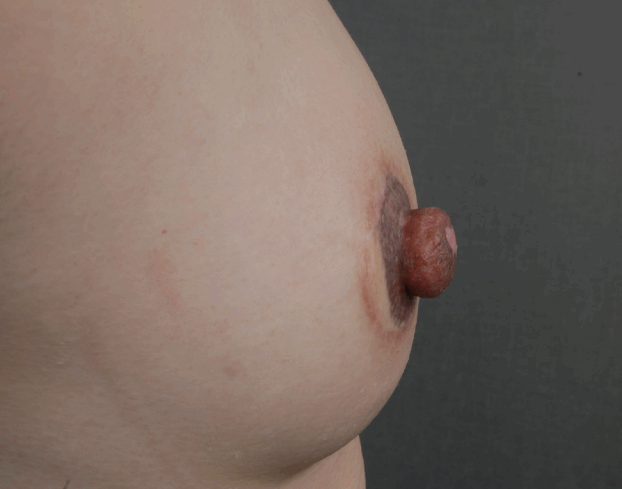
A line is traced circumferentially, marking the level of amputation on the donor nipple. It is 5–7 mm from the nipple tip when the nipple is in a flaccid state (Fig. 1). Lignocaine with adrenaline is injected sparingly to control bleeding and to create tumescence. The main challenge with cutting the nipple is its flaccidity and tendency to slip, coupled with the toughness of the nipple tissue resisting the passage of the blade. The result is often a scalloped cut with an irregular base. To eliminate this problem, the nipple is transfixed with a pair of Sims straight needles and cut with a pair of No. 11 blades, sweeping simultaneously outwards from the center (Fig. 2). Thus, an even cut is achieved (Fig. 3). Low-wattage bipolar cautery is applied over the donor site, followed by a tie-over alginate dressing to prevent rebound bleeding. The donor site is left to heal by secondary intention.
Nipple guillotine technique
A straight Sims needle is used for transfixing the tip of the nipple, The nipple graft is sutured in place with nylon 8-0 sutures. and 2 No. 11 blades are used for cutting outwards in tandem.
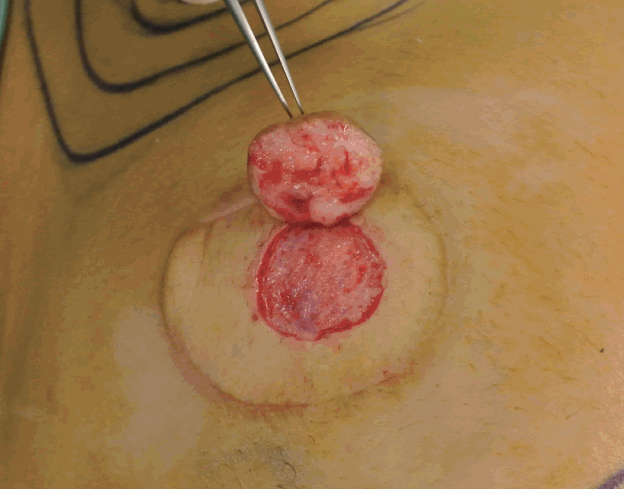
Nipple graft and the de-epithelialized recipient site
Nipple graft and the de-epithelialized recipient site.
At the recipient site, which is either the skin paddle of the transverse rectus abdominus myocutaneous flap or latissimus dorsi flap, a circle with a diameter of 1–1.5 cm is drawn according to the dimensions of the graft. The skin is thinly scored with a No. 15 blade and evenly de-epithelialized with a pair of Paget scissors to expose the subdermal plexus. Fat should not be exposed, as it is avascular. A well-prepared bed should have punctate bleeding. Local anesthesia is avoided at the recipient site, as it masks bleeding.
The nipple graft is then sutured to the recipient site with interrupted 8-0 nylon sutures under the operating microscope. Precise apposition of the skin edges is necessary to maximize dermal contact between the graft and its bed (Fig. 4). This step maximizes revascularization through the subdermal plexus. Stacked foam donut dressings (Allevyn, Smith & Nephew, United Kingdom) are used for protecting the nipple graft. Donut dressings are anchored with stitches to prevent displacement. Then, 1% tetracycline ointment is applied over the graft and the suture line, and the entire construct is left sealed for 7 days.
The patient is prescribed oral antibiotics and counselled that the nipple will be discolored for 3 weeks before pinking up. After the first inspection on postoperative day 5, dressings are changed in an atraumatic manner every 3 days by the surgeon in charge. Sutures are left in place for 4–6 weeks. The areola is created with tattooing or skin grafting depending on the patient’s preference.
Results
This guillotine technique was performed on 9 patients, with an average follow-up of 2.9 years (range, 1–4.5 years) (Table 1). The average graft height ranged from 4 mm to 8 mm and was maintained throughout the follow-up period. Our patients wore Allevyn splints for 6 months. One failure occurred due to graft dislodgement during dressing change. The outcome of the technique is illustrated in Figs. 5–7.
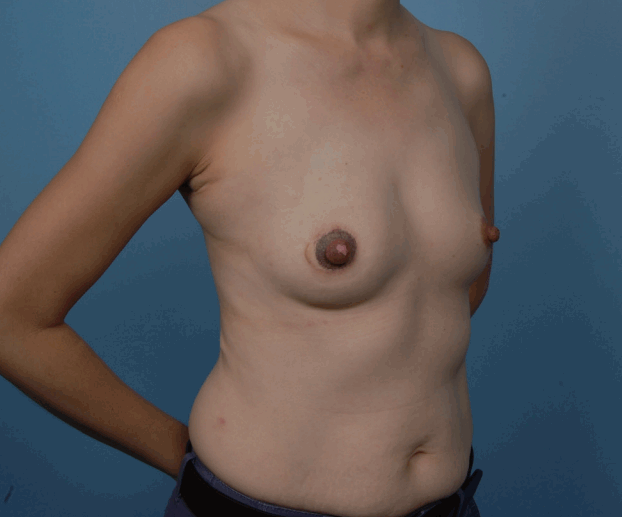
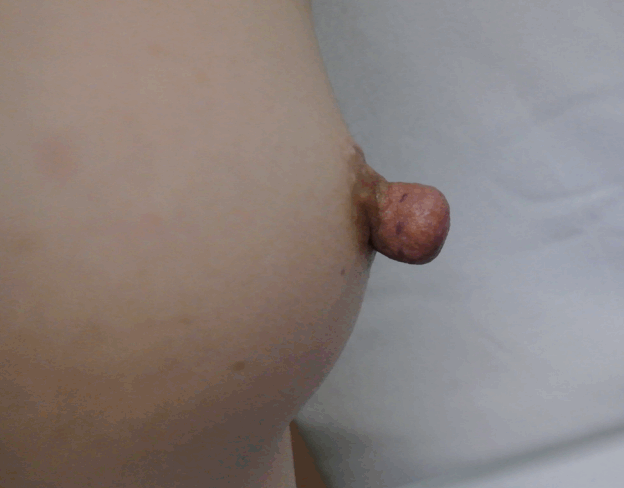
Nipple guillotine technique (right oblique view)
A 42-year-old woman underwent right breast reconstruction with a latissimus dorsi flap, followed by nipple reconstruction with the nipple-sharing method. The donor site healed well.
DISCUSSION
In 1950, Kiskadden described a case in which the surgeon, while taking split-skin grafts for burn coverage, inadvertently picked up the surface of a nipple and areola in his grafts. This graft took on the burned arm, while the donor site regenerated a reasonable nipple and areola. Inspired by this, Adams [10] and Millard [11] performed the first nipple-sharing procedure for nipple reconstruction, using a No. 10 blade to harvest the dome of the donor nipple. Nipple graft harvesting has since been done in various ways. Spear used a nylon traction suture to improve control on the nipple when cutting with a No. 11 blade [7]. Sakai and Taneda [3] harvested a rim of the nipple near its base and closed the denuded area by invaginating the nipple. The new nipple was reconstructed by spirally insetting the graft [3]. Other surgeons used a sagitally split nipple as a graft in cases where the nipple was flat [1,12].
Structurally, the nipple contains 15–20 lactiferous ducts, which are lined with luminal epithelial cells. Interspersed between these ducts is myoepithelial and stromal connective tissue, which is supported by a dense capillary network. With precise apposition, rapid revascularization can occur between the nipple base and the recipient site when their respective capillary networks link up. Plasmatic imbibition is the greatest at the base of the graft, and we believe that it progresses upward between the ducts by capillary action. As stromal vascular elements repopulate the graft, it becomes completely adherent to the bed. A meticulous microsurgical technique optimizes the conditions for the graft take because it allows healing from both the bed and the skin edges.
Detailed postoperative care was a key to the success of our two-blade guillotine technique. Initial dressing changes were performed avoiding any movement that might disrupt capillary inosculation. If the height of the graft exceeded 5 mm, hyperbaric oxygen therapy was instituted to increase local tissue oxygen tension. As for the donor site, the secondary healing involved minimal scarring, and we observed that ductal patency was maintained because the terminal openings of the ducts were not closed.
Notes
No potential conflict of interest relevant to this article was reported.
Notes
PATIENT CONSENT
The patient provided written informed consent for the publication and the use of their images.