Case of late hematoma after breast augmentation
Article information
Abstract
We present a case report of a patient who experienced a late, spontaneous breast hematoma 26 years after primary breast augmentation. Late hematomas are a rare complication of breast augmentation with uncertain etiology. In this case, there was no trauma, calcifications, or implant rupture. We believe the patient’s hematoma was secondary to erosion of a capsular vessel due to capsular contracture.
INTRODUCTION
Bleeding is a well-known and well-characterized acute complication following breast augmentation. This occurs in about 1% of patients [1-3]. Acute hematoma incidence is not significantly associated with type of implant surface texture or type of surgical procedure [2]. Conversely, late hematomas are a very rare complication of breast augmentation, documented by only 15–20 case reports. Here we describe a case in which capsular contracture appears to have contributed to the development of a late hematoma, in the absence of trauma, calcification, or implant rupture.
CASE
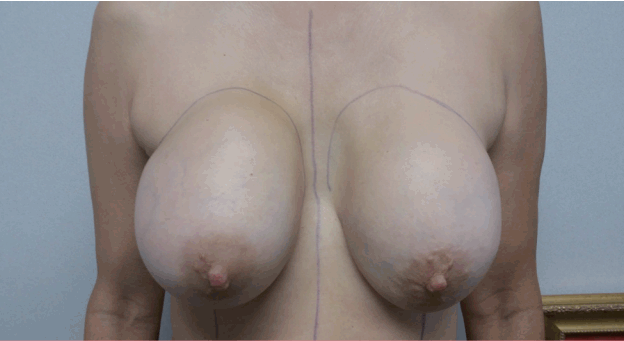
A 58-year-old woman, gravida 2 para 2, underwent a bilateral subglandular breast augmentation in August 1990 with polyurethane-coated, 325 mL silicone gel Replicon implants via inframammary incisions. The patient’s postoperative course was uneventful. The patient had soft breasts until 2011, when she began to experience capsular contractures bilaterally. A mammogram from 2015 suggested rupture of the right implant. In May 2016, the patient began to experience a gradual increase in size and distortion of the right breast, which was associated with mild pain. On physical examination, the patient had evidence of bilateral capsular contracture, Baker IV on the right and Baker III on the left. The right breast was firm and deformed (Figs. 1, 2). There were no palpable masses or lymphadenopathy.
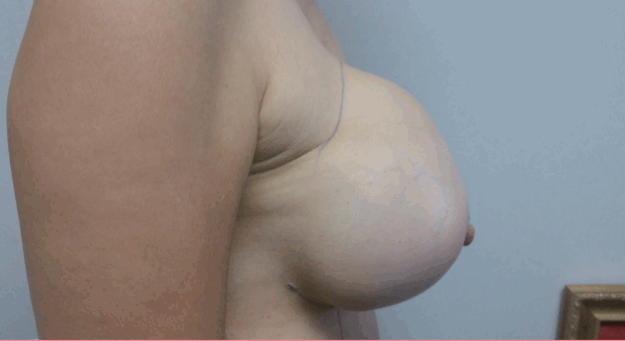
Front view of the hematoma
Front view of the hematoma shows the swelling of the upper pole of the right breast with mild bluish discoloration.
In May 2016, the patient underwent bilateral implant exchange with total capsulectomies. The capsule on the right was darkly pigmented and 150 mL of uncoagulated dark blood was evacuated from the capsule (Fig. 3). The implant was removed intact and total capsulectomy was performed. The capsule was strongly adherent to surrounding tissues and difficult to excise. There was an area of erosion in the medial aspect of the capsule as shown in Fig. 4. No calcifications were noted. The old implants were exchanged for 322 mL textured gel implants (Allergan).
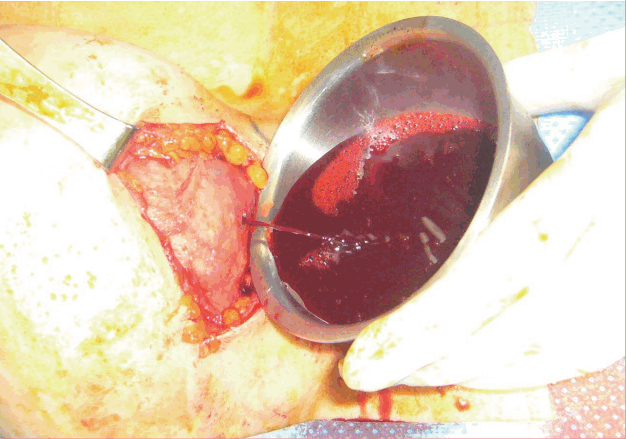
Right opened capsule
The right capsule has been opened with a stab incision, demonstrating outflow of uncoagulated blood.
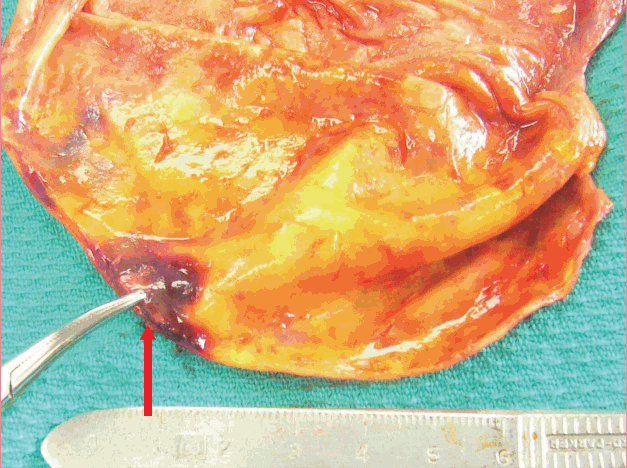
Right opened capsule from interior view
The right capsule is shown from the implant side. The red arrow is pointing to the area of vascular erosion and bleeding.
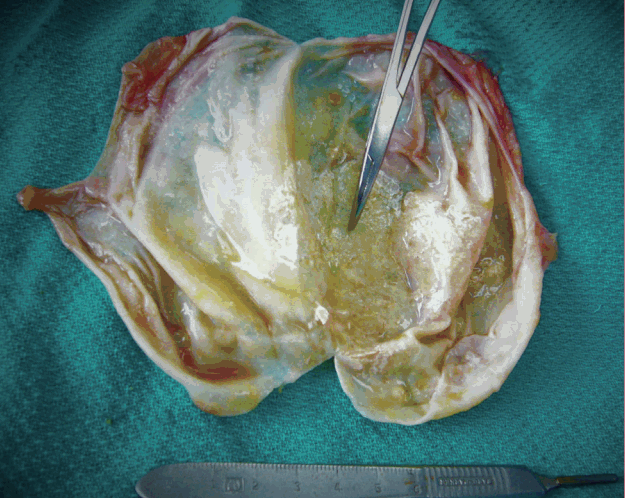
The capsule on the left was pale and no blood or fluid collections were noted. The implant was removed intact and total capsulectomy was performed without difficulty. The left capsule was barely adherent to the underlying pectoralis muscle and overlying breast tissue. It was easy to excise with minimal blood loss. The removed capsule, shown in Fig. 5, showed mild capsular calcifications primarily involving the anterior surface of the implant.
DISCUSSION
Late complications of breast augmentation with implants are varied, but fairly rare, including anaplastic large T-cell lymphoma, late seroma, and late hematoma. They often present as late progressive enlargement or deformation of the affected breast [4-7]. The late complications of breast augmentation with implants are varied, but fairly rare, including anaplastic large cell lymphoma, late seroma, and late hematoma. The etiology of late hematoma following breast augmentation has been poorly characterized due to its rarity. The first case was reported by Georgiade et al. [8] in 1979. They proposed that corticosteroid usage at time of placement was responsible for the erosion and subsequent hematoma. Currently, general consensus holds that late hematomas most likely result from erosion of capsular vessels due to trauma, inflammation, microfracture of the capsule, or friction of implant against the capsule [9-12]. In one case, trauma in the form of amorous squeezing was implicated [13]. In this case report, capsular contracture appears to have been the primary mechanism for the late hematoma. Previous case reports have suggested that polyurethane coating, as in this case, may provoke an intense inflammatory reaction which would subsequently place patients at higher risk for developing capsular contracture and, possibly, late hematoma [9]. However, it has since been shown that polyurethane-coating actually decreases the risk of capsular contracture development [14]. In this patient, gradual disintegration of the protective polyurethane coating may have contributed to the development of capsular contracture, culminating in late hematoma development 26 years later due to pressure against capsular vessels (Fig. 4).
In conclusion, polyurethane-coated implants have been discontinued in the United States since 1992, however, they are still present in a number of patients. Though polyurethane coating is protective against capsular contracture, as the coating ages and disintegrates, many more patients may begin presenting with complaints of capsular contracture. In rare cases, they may also present with late hematoma likely due to capsular vascular erosion.
Notes
No potential conflict of interest relevant to this article was reported.
Notes
Ethical approval
The study was performed in accordance with the principles of the Declaration of Helsinki. Written informed consents were obtained.
Patient consent
The patients provided written informed consent for the publication and the use of their images.