INTRODUCTION
Up to the present, autologous skin grafts or flaps have been widely used for repairing skin and soft tissue defects. However, improved reconstruction poses greater donor site problems. To resolve these problems, skin substitutes have been developed using tissue engineering [
1]. However, in order for skin substitutes to be applied to patients with full-thickness skin loss, the substitutes should function as an alternative to autologous skin, form an effective barrier against bacterial invasion, minimize inflammation and scar formation, improve fibrovascular tissue ingrowth, and have excellent reproducibility [
2,
3].
A dermal substitute, which is the most extensively developed of the current skin substitutes, consists of a collagen scaffold that comprises mainly collagen containing either dermis extracellular matrix or cultured fibroblasts. A bilayered skin substitute that contains epidermis-forming keratinocytes attached on a collagen scaffold with cultured fibroblasts is ideal. However, its take rate is problematic.
We have generated a novel dermal substitute containing a collagen scaffold by combining chondroitin-6-sulphate (C6S) with atelocollagen (ATC) extracted from calf dermis. To lessen the antigenicity of ATC, the telopeptides located at the terminal of the collagen molecule were removed using pepsin. C6S was added to increase stability and the scaffold was treated by physical cross-linking in a dehydrothermal (DHT) treatment alone or with an additional chemical cross-linking involving 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC). We used an amniotic membrane (AM) instead of silicone membranes that temporarily served as epidermis, to protect the wound from the external environment, to keep the wound moist, and to aid in the promotion of wound healing through various growth factors.
This study was undertaken to generate a new dermal substitute and to compare it with previously designed dermal substitutes, in order to confirm its usefulness in clinical practice through animal experiments.
DISCUSSION
Dermal substitutes should be physically strong and easy to handle, endure contraction through durability, continue to attach viable fibroblasts during the whole wound healing process, enable the reconstruction of the dermal lattice with fibroblasts, and should endure hydrolysis during their presence in the wound until they are finally absorbed after complete wound healing [
2,
5].
The use of a collagen scaffold as a dermal substitute reduces wound contraction and scar formation, and promotes epithelialization. Collagen has attracted much attention because it stimulates wound healing through its strong affinity to the soft tissue and blood vessels, and it can be replaced with host cells and vessels through acceleration of their penetration into the scaffold [
6]. Because chemical and physical changes in collagen may affect the recipient tissue morphologically and physiologically, we attempted to investigate whether changes in collagen would affect its affinity to host cells.
In this study, the novel substitute displayed many advantages over substitutes currently in use. We used the AM instead of a silicone membrane to obtain the physical protection barrier functions as well as the anatomical and physiological functions similar to epidermis. The AM consists of three layers: the epithelial layer, basement membrane, and mesenchymal layer. The epithelial layer consists of a single layer of epithelial cells, and the basement membrane is a thin membrane composed of reticular fibers that attach to the epithelial layer by interdigitation. The mesenchymal layer is an avascular collagen matrix that is divided into compact, fibroblast, and sponge layers [
7,
8]. Although the AM has some variations in structure, it has histologically similar structures to the skin composed of the multilayered epithelium and basement membrane and, thus, has advantages in wound healing such as promotion of epithelialization, reduction in inflammatory reactions, and inhibition of scar formation and bacterial growth [
9,
10]. Promotion of epithelialization using AM has been clinically utilized for various skin lesions such as burns and ulcerations, and many growth factors have been identified in the AM by experimental studies. Koizumi et al. [
11] documented that the AM expresses mRNAs of 8 growth factors that remarkably influence epithelial regeneration. Hao et al. [
12] demonstrated that the AM shows mRNA expression of the interleukin-1 receptor antagonist and interleukin-10. In addition, Tseng et al. [
13] suggested that inflammatory reactions are significantly reduced in the cornea, and limbus grown on the AM, probably due to reduced mRNA expression of transforming growth factor (TGF) receptor II and TGF-β-1, 2, and 3 through the inhibition of the TGF-β-signaling system. Inhibition of the TGF-β signaling system can reduce scar formation through the activation of fibroblasts into myofibroblasts. It has been reported that the amnion and chorion have an antibacterial effect against hemolytic
Streptococcus group A,
Escherichia coli, and
Pseudomonas [
14].
AM has been used as a temporary substitute because it protects skin wounds from the external environment, it decreases the loss of water, electrolytes, protein, and energy, it reduces pain, it compensates for the disadvantage of dermal substitutes in current use that are prone to infection, and it promotes wound healing through various growth factors [
15].
We generated collagen scaffolds by combining C6S with atelocollagen extracted from calf dermis. To lessen the antigenicity of the atelocollagen, the telopeptides located at the terminal of the collagen molecule were removed using pepsin. In this atelocollagen, the percentage of tyrosine in relationship to the total amino acids was 0.12%, which was significantly lower than that of the criterion for non-antigenicity of collagen (0.2%), suggesting that inflammatory reactions can be adequately lessened when grafted in human skin. C6S is a high-molecular substance that inhibits proteolytic enzymes, plays a crucial role in the binding and regulation of growth factors, and influences chemotaxis, migration, hyperplasia, and differentiation of cells [
16]. Thus, the dermal substitute we generated by the addition of C6S could reduce biodegradation of collagen caused by proteolytic enzymes, and could lead to easy migration of cells from the surrounding tissue into this collagen scaffold due to the increased elasticity, durability, and porosity compared to a collagen scaffold alone [
17].
DHT/chemical cross-linking inhibits the rapid degradation of a collagen scaffold, lessens its antigenicity, and enforces its physical properties when it is grafted on human skin. A DHT cross-linked collagen scaffold is more stable, maintains the same porosity as that prior to the treatment without distortion, and does not induce cytotoxic substances. This collagen scaffold is distorted in an EDC solution due to hydrophobicity induced by the initial DHT-treatment. Although this phenomenon can be prevented to some extent when crosslinking is conducted in the presence of ethanol, such chemical substances can induce cytotoxic substances
in vivo. However, an EDC-treated collagen scaffold can prevent rapid degradation because it becomes resistant to proteolytic enzymes [
16].
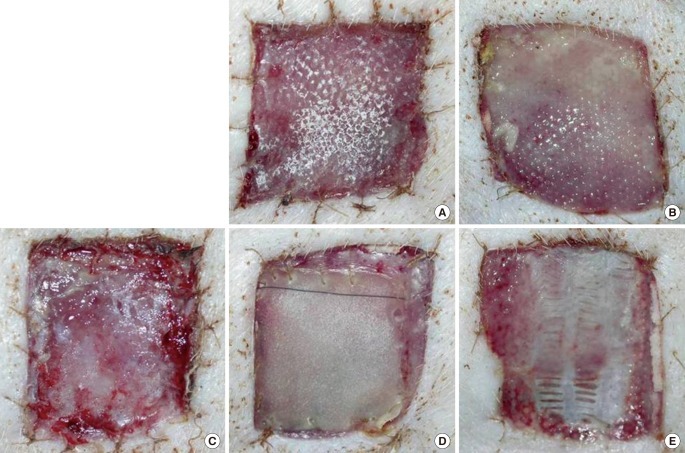
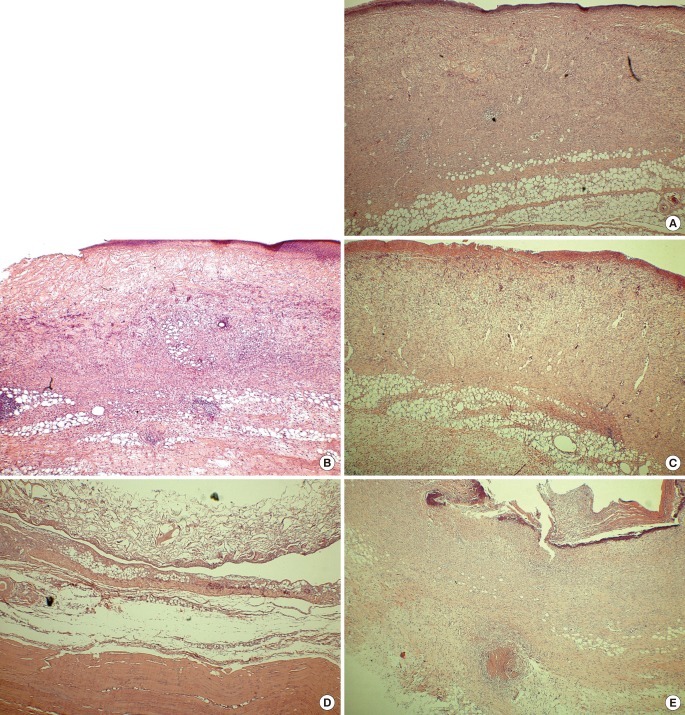
In this study, the results were significantly different between the experimental groups. Groups 1 and 2 (the DHT-treated groups) showed a more rapid wound healing than the other groups, suggesting that DHT-treated collagen scaffolds have a strong affinity to cellular components of the surrounding soft tissue and blood vessels, which was evident from the electron microscopic findings (
Supplemental Fig. 3). The results support the idea that it is conceivable that atelocollagen may inhibit inflammatory reactions by lessening antigenicity, whereas C6S may promote wound healing by reducing biodegradation caused by proteolytic enzymes, which increases physical stability and induces the ready migration of cells from the surrounding tissue into the collagen scaffold through its increased porosity. In addition, AM used as a temporary protection membrane can promote wound healing, probably through the secretion of various growth factors.
In conclusion, our atelocollagen dermal substitute that was generated using atelocollagen, AM, and C6S may be more reliable than collagen dermal substitutes in current use.