DISCUSSION
The human axilla produces a characteristic odor called an "axillary odor." In the axilla, various compounds are secreted by many types of glands, such as apocrine, eccrine, and sebaceous glands. The axillary odor is caused by interactions between apocrine secretions and the normal flora of the axilla.
There have been numerous studies on axillary odor in terms of chemistry, gender differences, intensity, and related microorganisms. In addition, ε-3-methyl 2-hexenoic acid has been identified as an axillary odor-related compound, which is secreted by the axillary apocrine glands in Caucasians. Akutsu et al. [
1] analyzed ε-3-methyl 2-hexenoic acid from individually collected axillary sweat and reported that ε-3-methyl 2-hexenoic acid is also found in the axillary sweat of Asians. Rennie et al. [
3] demonstrated that
Corynebacterium species produce odor in the axillary region and that the axillary microflora is specifically involved in underarm odor production.
Numerous treatment methods for axillary osmidrosis have been introduced. Hurley et al. [
4] reported en
bloc excision of the axillary skin and soft tissue. Inaba et al. [
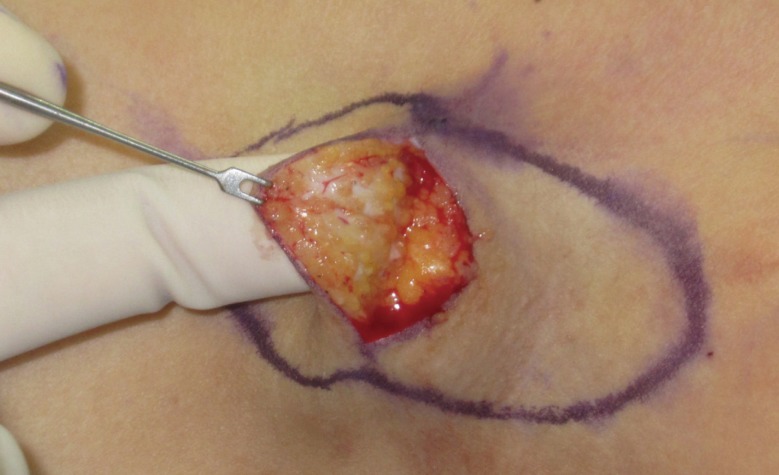
5] introduced selective surgical excision of the apocrine and eccrine glands, which has become one of the most popular surgical methods. We called this surgical method "conventional subdermal shaving" in our study. Alternatively, suction curettage with mechanical liposuction cannulas or ultrasonic suction instruments is also a well known method. Recently, many centers have reported methods for treating axillary osmidrosis with lasers, including CO
2 and Nd-Yag lasers [
6]; however, conventional subdermal shaving is still the treatment of choice [
2]. Previous studies have reported complications of the subdermal shaving method, including hematoma, seroma, skin flap necrosis, wound infection, and the recurrence of malodor [
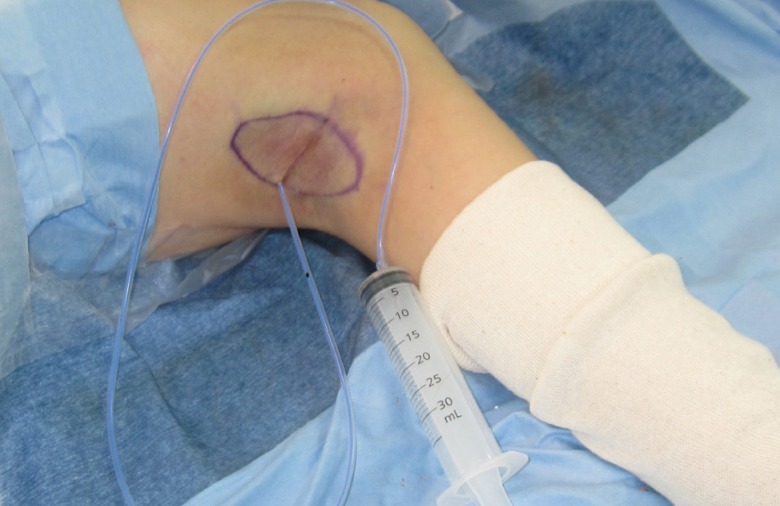
2]. These complications are caused by failure to control microbleeding, subdermal plexus injury due to aggressive shaving, contamination with the normal flora, and remnant apocrine glands. Like a double-edged sword, aggressive shaving for the removal of apocrine glands can increase the possibility of subdermal plexus injury, while minimal shaving in order to avoid subdermal plexus injury can lead to recurrence of osmidrosis due to remnant apocrine glands. To avoid these complications, alternative options, such as the use of tumescent solution and tie-over compression [
7] have been introduced. Many authors have reported performing these alternative methods to avoid the possible complications mentioned above.
The chemical, physiologic, biochemical nature of sclerosants deforms the target tissue, whose byproducts induce cell damage, fibroblast proliferation, and sclerosis. In addition, thrombosis, protein extraction from lipids, protein denaturation, cell dehydration, and physical obstruction by polymerization impair functions of the target tissue [
8]. Ethanol was first described as a sclerosant in a canine renal model in the 1980s [
9]. Its mechanisms of action include the denaturation and extraction of surface proteins, cell dehydration, coagulation, thrombosis, and fibrinoid necrosis [
10-
12]. Its effectiveness is related to the concentration, exposure time, and injection speed [
13].
Sclerotherapy using ethanol has been widely used in the treatment of esophageal piles, vascular malformations, varicose veins, and benign cysts, such as thyroglossal cysts, renal cysts, and ovarian endometriomas. It has recently been used for the treatment of chronic recurrent ischial pressure sores following surgical treatment [
14] and rheumatoid bursal cysts [
15]. Absolute ethanol causes damage to the endothelial cells in abnormal vessels, sterilizes the environment by its bactericidal effect, and alters the subcutaneous layer by coagulation as well as tissue modification of cell proteins. Since absolute ethanol causes tissue conglutination and obliteration of the dead space, it has been widely used to treat cavitary disease. Systemic side effects are rare unless absolute ethanol is directly injected into blood vessels [
14]. Absolute ethanol has rarely been reported as a cause of hypersensitivity. In a previous study, absolute alcohol skin prick test results were negative in all patients who showed allergic reactions after alcohol intake [
16].
Our surgical procedure minimized subdermal plexus injury by using less aggressive shaving than the conventional method. For this reason, the frequency of complications, such as skin necrosis, desquamation, and wound disruption, decreased. Discharge from the operation site also decreased and resulted in earlier drain removal by 1.09 days than the conventional method. Complications, such as hematoma and seroma, were not seen in the study group. Postoperative satisfaction was similar in the study and control groups. Other late complications, including recurrence of osmidrosis, hypertrophic scar formation, and limitation of shoulder movement, did not occur in the study group over a 6-month follow-up period.
Sclerotherapy combined with minimal subdermal shaving may be useful for treating axillary osmidrosis through several mechanisms. First, absolute ethanol can reduce the incidence of seroma and hematoma by coagulation, cell modification, and obliteration of dead space. Furthermore, it allows the skin flap to adhere to its recipient site. Second, as a result of its cytotoxicity, unrecognized remnant apocrine glands can be degraded and ablated. Because of its apocrine gland ablation effect, surgeons do not need to perform excessive surgical removal of the subdermal layer, leading to preservation of the subdermal plexus, which allows for better skin flap survival. Additionally, owing to its bactericidal effect, absolute ethanol can eliminate causative organisms which colonize the normal axillary skin. Lastly, the sclerotherapy is relatively simple and requires a shorter operation time than the conventional method. As a result, a low complication rate and early recovery following earlier drain removal was shown.
This study has some limitations. First, the small number of cases limits statistical analysis. Second, the severity of osmidrosis could differ between the two axillae before surgery, which may be a confounding factor. Third, follow-up longer than 2 years was not feasible, and therefore we were not able to check for scar tissue formation or late recurrence. Fourth, we could not perform histopathologic studies because it was difficult to obtain consent from each patient, and the wounds healed well without any complications. Despite these limitations, our method may be effective in treating patients with axillary odors, and was easy to perform with a low complication rate.