METHODS
ACS-NSQIP
Data is extracted from patient medical records, physician office records, and telephone interviews by trained surgical clinical nurse reviewers (SCNRs). All information is subsequently deidentified [
11].
Ethical approval
De-identified patient information is freely available to all institutional members who comply with the ACS-NSQIP Data Use Agreement. The Data Use Agreement implements the protections afforded by the Health Insurance Portability and Accountability Act of 1996 and the ACS-NSQIP Hospital Participation Agreement. This study conforms to the Helsinki Declaration. The ACS-NSQIP and the hospitals participating in the ACS-NSQIP are the source of the data used herein; they have not verified and are not responsible for the statistical validity of the data analysis or the conclusions derived by the authors.
Patient population
We retrospectively reviewed database participant files from 2006-2010 for all cases with 'plastics' recorded as the primary service. At total of 15,289 plastic surgery cases were identified. No cases within this cohort were without proper wound classification designation, and no patients died on the same day of surgery.
Outcomes
The outcomes of interest in this study were SSIs, classified as superficial, deep, or organ/space, overall complications, reoperation, and mortality. An SSI was defined in the ACS-NSQIP database, based on CDC criteria, as an infection within 30 days of the primary operation that had purulent drainage with or without laboratory confirmation, organisms isolated from an aseptically obtained culture, and/or a diagnosis by the attending surgeon. Identification of an abscess indicated a deep or organ/space SSI. Additionally, at least one of the following signs or symptoms must have been present: pain or tenderness, localized swelling, redness, or warmth, an incision deliberately opened by the attending surgeon, or in the case of deep SSI, fever >38℃. The depth and location of the infection determined the type of SSI. A superficial SSI involved only the skin or subcutaneous tissues of the incision; a deep SSI involved the muscle and fascial layers; an organ/space SSI involved any part of the anatomy. An infection involving both the superficial and deep tissues was recorded as a deep SSI, as was an organ/space SSI that drained through the incision [
12,
13].
Overall SSI was defined as ≥1 superficial SSI, deep SSI, or organ/space SSI. Overall complication was defined as having ≥1 of the following ACS-NSQIP postoperative adverse events: superficial SSI, deep SSI, organ/space SSI, wound disruption/dehiscence, pneumonia, unplanned intubation, pulmonary embolism, failure to wean from ventilator, renal insufficiency, progressive renal failure, urinary tract infection, stroke, coma, peripheral neurologic deficiency, cardiac arrest, myocardial infarction, bleeding requiring a transfusion, deep venous thrombosis (DVT), and sepsis/septic shock. All complication, reoperation, and mortality rates were tracked for 30 days postoperatively.
Risk-adjustment factors
Patient demographics and medical comorbidities were tracked as potential confounders. Demographic data collected included age, gender, and race. Medical comorbidities included obesity (body mass index [BMI]≥30), diabetes, dyspnea, ascites, renal disease, chronic obstructive pulmonary disease (COPD), current pneumonia, ventilator dependence, chronic steroid use, bleeding disorders, heart failure, myocardial infarction within 6 months of operation, peripheral vascular disease, disseminated cancer, weight loss of >10% body weight within 6 months of operation, current chemotherapy or radiotherapy, neurologic deficit, preoperative transfusion, and preoperative sepsis. Alcohol use, defined as >2 drinks per day, and smoking status were tracked as behavioral risk factors. Additionally, operative time, a recognized risk factor for SSI, was included in the risk adjustment.
Statistical analysis
Patient demographics, risk factors, and postoperative outcomes were compared using chi-square tests for categorical variables and independent t-tests for continuous variables, with significance set at P≤0.05.
Multivariable logistic regression analysis was conducted to examine the effect of wound classification on 30-day postoperative surgical site infections, overall complications, reoperation, and mortality. Potential confounding risk factors were determined using a bivariate screen that identified variables with significance at P≤0.20. All data analyses were performed using SPSS ver. 20.0 (IBM Corp., Armonk, NY, USA).
RESULTS
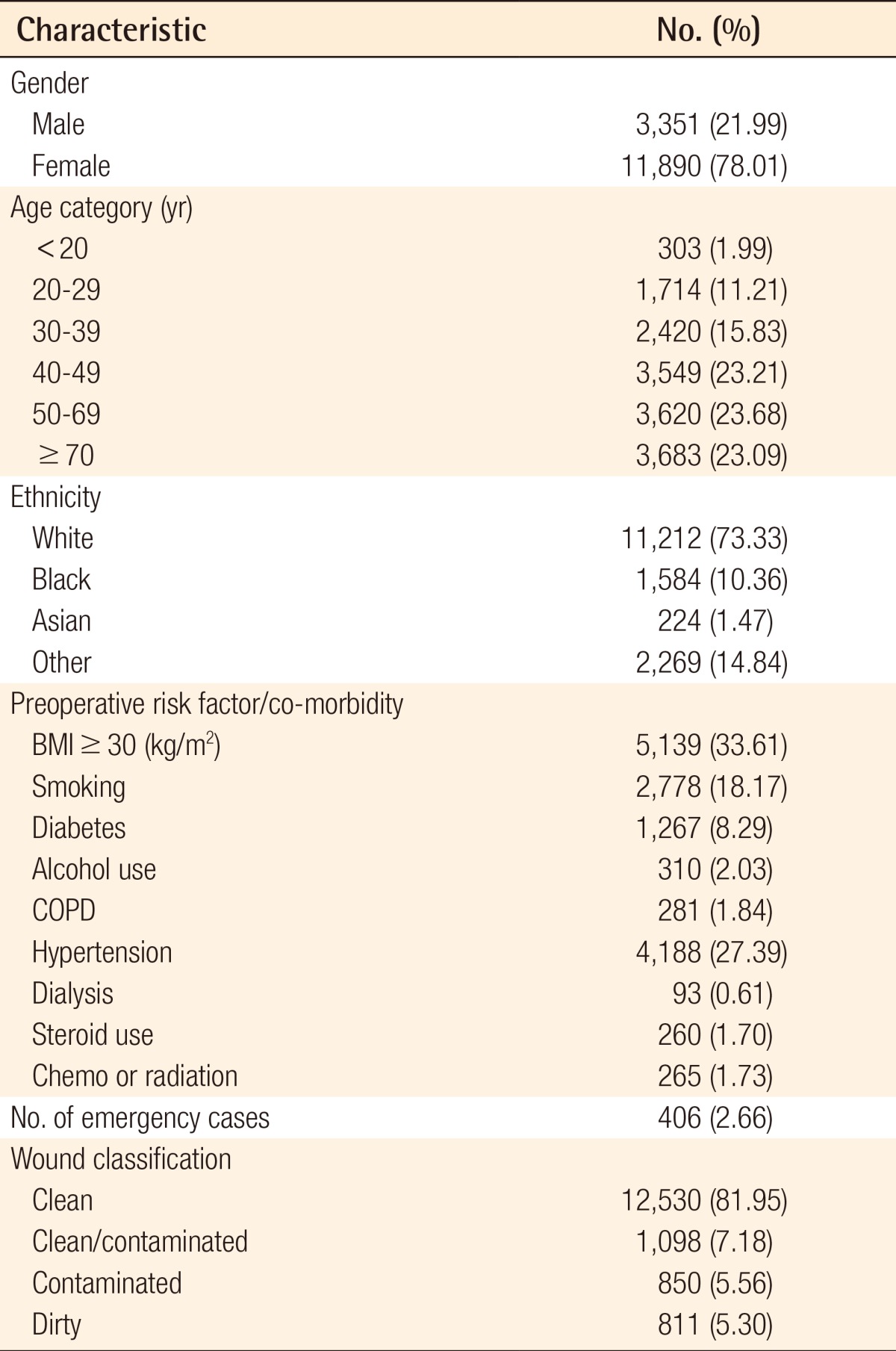
A total of 15,289 plastic surgery cases were extracted from the database. No cases were excluded. From this cohort, 81.95% of the procedures were classified as clean, 7.18% clean/contaminated, 5.56% contaminated, and 5.30% dirty (
Table 2). Over three-fourths of the patients were female, and nearly the same proportion was Caucasian. The average age of the group was 48.36 years, with equal distribution between the 40 to 49, 50 to 59, and ≥60 year old age brackets. The most common comorbidity was obesity at 33.61%, followed by diabetes at 8.29%. The two most common preoperative risk factors were hypertension (27.39%) and smoking (18.17%). Four-hundred and six of the total cases (2.66%) were classified as emergency cases. When comparing patients with and without recorded surgical site infections, there were notable differences in obesity (53.69% vs. 33.68%, P<0.001), diabetes (15.16% vs. 8.08%, P<0.001) and hypertension (37.58% vs. 27.09%, P<0.001). There were also significant differences in the proportion of patients with a history of COPD (3.74% vs. 1.78%, P=0.001) and on dialysis (1.76% vs. 0.57%, P=0.001), although the overall rates were low.
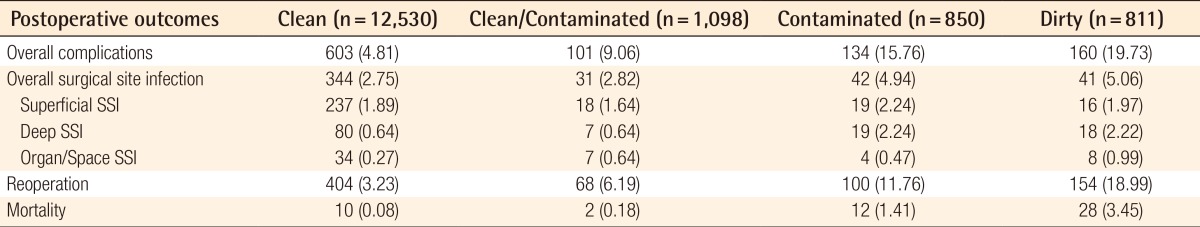
There were 998 cases with a postoperative complication, representing 6.53% of the total plastic surgery procedures analyzed (
Table 3). Nearly one-fifth of dirty procedures incurred a post operative complication, compared to 15.76% of contaminated cases, 9.20% of clean/contaminated, and 4.81% of clean cases (
Table 3). Surgical site infections (SSIs) made up the majority of these postoperative complications, occurring in 458 cases (3.00%). Reoperation and mortality rates also increased as the wound classification transitioned from clean to dirty (
Table 3).
Overall, 1.89% of plastic surgery patients suffered a superficial SSI, 0.81% experienced a deep SSI, and 0.35% had an organ/space SSI. Superficial SSIs occurred at similar rates across wound classification cohorts: 1.89% of clean cases, 1.64% of clean/contaminated cases, 2.24% of contaminated cases, and 1.97% of dirty cases. There were similar rates of occurrence for deep SSIs in the clean and clean/contaminated groups (0.64%), while 2.24% of contaminated cases and 2.22% of the dirty cases experienced a deep SSI. Organ/space SSI occurred in less than 1% of each wound classification cohort (
Tables 3,
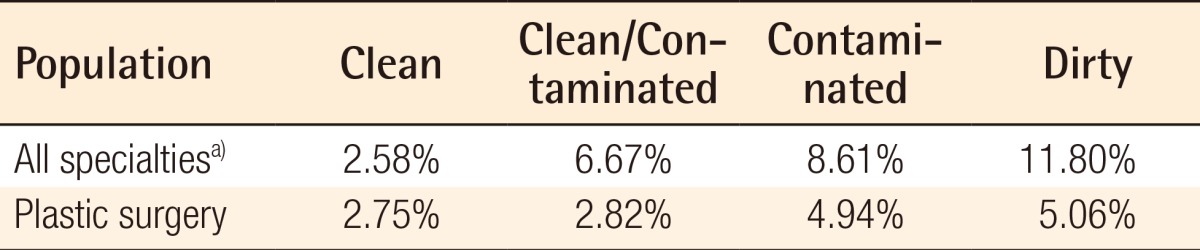
4). When compared to all other specialties, plastic surgery had lower rates of surgical site infections in clean/contaminated, contaminated, and dirty cases (
Table 4).
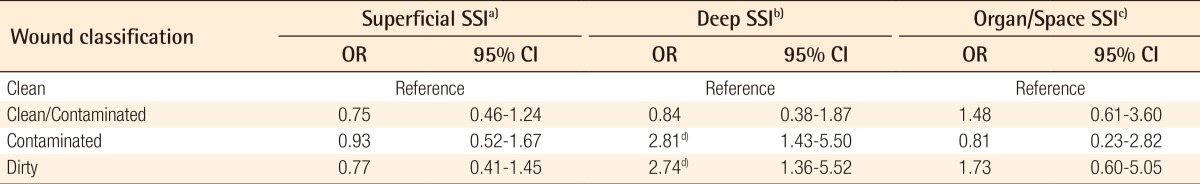
Adjusted odds ratios (OR) and 95% confidence intervals (CI) for superficial, deep, and organ/space SSIs are listed in
Table 5. With clean operations as a reference group, clean/contaminated, contaminated, and dirty cases had a statistically similar risk of a superficial SSI. The odds ratios for deep SSIs were 0.84 (95% CI, 0.38-1.87; P=0.666), 2.81 (95% CI, 1.43-5.50; P=0.003), and 2.74 (95% CI, 1.36-5.52; P=0.005) for clean/contaminated, contaminated, and dirty cases, respectively. Wound classification did not appear to be a significant predictor of an organ/space SSI; the odds ratios for an organ/space SSI were 1.48 (95% CI, 0.61-3.60; P=0.399), 0.81 (95% CI, 0.23-2.82, P=0.731), and 1.73 (95% CI, 0.60-5.05; P=0.322) for clean/contaminated, contaminated, and dirty procedures, respectively.
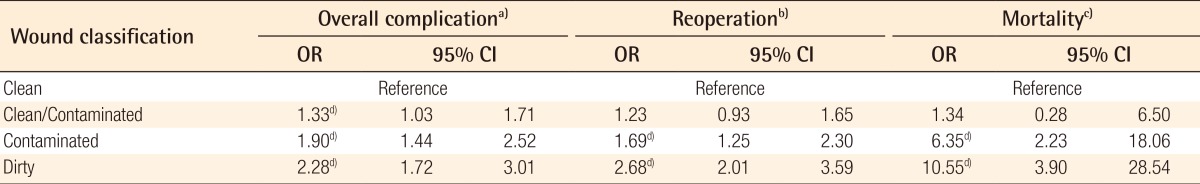
Regression analysis was also conducted on our remaining outcomes of interest-overall complications, reoperation, and overall mortality (
Table 6). Clean/contaminated procedures were comparable to clean cases in their risk of reoperation and mortality. However, clean/contaminated procedures increased the risk of overall complications by 33% (adjusted OR, 1.33; 95% CI, 1.03-1.71; P<0.001). Contaminated cases were associated with adjusted ORs of 1.90 for overall complications (95% CI, 1.44-2.52; P<0.001), 1.69 for reoperation (95% CI, 1.25-2.30; P=0.001), and 6.35 for 30-day mortality (95% CI, 2.23-18.06; P=0.001). Dirty cases had a 2.28 greater odds of having a postoperative complication (95% CI, 1.72-3.01; P<0.001), a 2.68 greater odds of reoperation (95% CI, 2.01-3.59; P<0.001), and a 10.55 greater odds of 30-day mortality (95% CI, 3.90-28.54; P<0.001) when compared to clean cases
DISCUSSION
This study is the first to investigate surgical site infections in plastic surgery by wound classification using a nationally validated surgical outcomes database. Overall, surgical site infection rates for plastic surgery were lower in non-clean, i.e. clean/contaminated, contaminated, and dirty, cases than the most recently reported rates for all surgical specialties represented by ACS [
14]. Additionally, our infection rates by wound class were universally lower than rates previously reported in a prospective study on plastic surgery cases [
15]. Such findings may serve as a benchmark for future outcomes studies. Over 80% of plastic surgery procedures were classified as clean, compared to approximately 50% of procedures across all ACS specialties [
14]. These numbers reflect a selection bias in plastic surgery with respect to surgical site infections and a tendency of plastic surgery operations to focus on the soft tissues.
Plastic surgery is acknowledged as a diverse specialty, with procedures addressing a variety of anatomic locations and representing a range of technical complexity. A recent literature review found the rate of SSIs in plastic surgery to vary greatly with the type of surgery performed: 0.001% to 7% in breast augmentation, 1.1 to 22% in breast reductions, 0.2% to 32.6% in abdominoplasty, and only 0% to 0.3% in rhytidectomy and less than 0.1% in browlift [
16]. Three of the four most common clean procedures captured in our study were focused on the breast, with mammaplasty being the most prevalent (19.80%).
In contrast, over 36% of dirty cases were pressure sore and flap procedures. Our total SSI rate of 3.00% (458 of 15,289 patients) is consistent with published reports in the plastic surgery literature [
16-
18]. As a finding from a multi-center database, our data regarding surgical infection rates may supersede the implied biases of individual surgical series based on only a few surgeons or institutions, as seen in previous studies. A prospective study conducted by Andenaes et al. [
15] found that wound infection rates escalated from 10.2% in clean plastic surgery cases to 37.5% in dirty plastic surgery cases. Our overall SSI rates were much lower-ranging from 2.74% in clean procedures to 5.06% in dirty operations. The discrepancy in infection rates is most likely attributable to the fact that our study utilized a more stringent definition of infection, based on CDC criteria; the study by Andenaes et al. [
15] employed a Wound Infection Score (WIS) system that included a single presentation of "edema," "redness," or "increased pain" in their definition of wound infection. Additionally, a learning curve may exist, with plastic surgeons being more selective when choosing patients for specific operations at the present time compared to the years captured by the Andenaes study.
Our results showed no observable incremental increase in infection rates when progressing from clean to dirty cases. While there does appear to be a slight correlation between wound classification and overall SSI, the relationship breaks down when stratified by type of infection. The highest rate of organ/space surgical site infections was associated with dirty procedures; however, the highest rates of superficial and deep SSIs occurred in contaminated operations. Our risk-adjusted multivariable regression model revealed that wound classification was not a significant predictor for two out of the three types of surgical site infections, namely superficial SSIs and organ/space SSIs. Contaminated and dirty wound categories were significant independent predictors of a deep SSI, associated with a nearly threefold increased risk of a deep SSI. These findings are in contrast to the study by Ortega et al. [
14], which analyzed infection rates by wound class across all surgical specialties captured in NSQIP. They demonstrated an increased rate of infection when advancing from clean to dirty procedures and found wound classification to be a significant predictor of superficial, deep, and organ/space surgical site infections by multivariate analyses.
The reasoning for the lower infection rates in non-clean plastic surgery cases compared to similar cases in the Ortega study is multifaceted. Patient selection is a presumed contributor, with plastic surgeons operating on individuals who may be at a baseline lower risk for infections. Additionally, the majority of surgical site infections were classified as superficial and thus involved only skin or subcutaneous tissue. Plastic surgeons are trained in procedures involving soft tissues and therefore might have invested more effort into techniques such as debridement of contaminated or devitalized tissues, dead space reduction, and meticulous layered closure to reduce the likelihood of complications in the superficial and deep tissue layers. The discrepancies in infection rates by wound class could also be due in part to the differences in the procedure characteristics that define the nonclean classifications. In the majority of non-clean procedures in the Ortega study, the alimentary tract was entered. Plastic surgeons only occasionally enter the alimentary or respiratory tracts, so the majority of their non-clean procedures are likely to be those associated with open wounds or trauma. Moreover, these non-clean procedures may involve surgical debridement or preparation of the surgical bed as the first step of the reconstructive operation. Thus, the operation is a treatment for the contaminated wound rather than the contaminated wound being a consequence of the procedure.
Although statistical analysis did not support wound classification as a predictor of most types of surgical site infections, it was proven to be significant in predicting overall complications. In acknowledgement of the baseline discrepancies in patients with and without recorded surgical site infections, variables that reached significance on bivariate screening were included in the regression analysis. Clean/contaminated cases showed a 1.33 increased odds of having a postoperative complication (95% CI, 1.03-1.71; P=0.027), contaminated cases held a 1.9 increased odds of having a postoperative complication (95% CI, 1.44-2.52; P<0.001), and dirty cases had a 2.28 increased odds of having a complication (95% CI, 1.72-3.01; P<0.001). It has been demonstrated that contaminated wounds containing greater than 105 bacteria per gram of tissue are not effectively treated by skin grafting, primary closure, or random cutaneous flaps. It has further been shown that in heavily contaminated wounds containing greater than 106 bacteria per gram of tissue, musculocutaneous flaps will dehisce [
19]. Such flap and graft failures may have been tracked as complications other than surgical site infections by NSQIP.
Wound classification was also a significant predictor of reoperation. Contaminated operations had a 69% increased risk for reoperation, and dirty procedures had over a 100% increased risk for reoperation. Additionally, contaminated and dirty cases were significant predictors for mortality, with contaminated procedures having an OR of 6.35 and dirty procedures having an OR of 10.55. While NSQIP does not track the reason for reoperation or cause of death, these results may reflect the possibility of increased microbial contamination contributing to instances that lead to reoperation or death, such as wound break-down and sepsis. Moreover, it may be that patients in a wound classification with a higher bacterial burden have preoperative characteristics and comorbidities that place them at increased risk for such adverse events.
The wound classification system was created to have universal utility in identifying procedures with an inherently higher risk of postoperative infection. However, the large majority of plastic surgery cases are clean cases, and the definitions of the other classifications as they pertain to plastic surgery are less clear. One single institutional study of wound classification found 19% of their cases to be misclassified [
20]. When the ACS-NSQIP data was narrowed to study plastic surgery alone, wound classification lost much if its significance as a predictor for surgical site infection. The classification scheme does not consider basic plastic surgical principles such as the inherent vascularity of the surgical site or the use of prosthetic material. A modified wound classification system may be necessary to strengthen its applicability to specific fields and help further standardize preoperative antibiotic regimens and surgical advancements.
Despite suggestions that the nearly 50-year-old classification system should be revised, no such modifications have been adopted [
21,
22]. However, Culver et al. [
9] did devise the NNIS SSI risk index, which did not address the wound classification scheme itself, but combined it with other known risk factors to improve predictive value. The NNIS SSI risk index assigned point values to the following three criteria: American Society of Anesthesiologists score of 3 or greater, wound classification of either contaminated or dirty, and procedure-specific excessive operative time. The risk index proved to be a better predictor of SSIs than wound classification alone. Optimally, however, a wound classification system could serve as a tool for risk-adjustment specific to the wound characteristics, e.g., size, location, depth, approach, and level of contamination, and would be independent of patient and other operative variables. We therefore suggest that any future modification of the wound classification system consider such focused variables as anticipated incision size, detailed anatomic locations, and surgical approach (laparoscopic versus open) in addition to the level of contamination to better predict infections.
Despite the statistical power of this multi-institutional database, there are limitations to this study. First, the database tracks only 30-day postoperative outcomes and cannot properly capture a second stage reconstructive procedure. A focus on 30-day outcomes eliminates the possibility of tracking implant infections defined up to 1 year after the initial operation. Furthermore, data is recorded once every eight days, which may have impacted the proportion of plastic surgery operations captured in our analysis. Additionally, the database has limitations regarding variables germane to plastic and reconstructive surgery. In particular, the rate of seroma, a common postoperative complication, is absent, which therefore reduces the attributable complication rate. Particularly pertinent to this study is the lack of preoperative and postoperative care variables, specifically regarding the use of antibiotics and drains. Although nothing specific is captured, it is assumed that every institution will have some form of antibiotic prophylaxis in place. As the database is revised on an annual basis, it is our hope that the aforementioned untracked variables are incorporated to further enhance future studies.