INTRODUCTION
Growth factors provide key bioactive cues
in vivo and are important for tissue engineering strategies. However, their short half-lives and uncontrolled dose-related adverse effects present challenges [
1]. Attempts have been made to incorporate growth factor delivery into scaffolds [
1]. Transforming growth factor beta 1 (TGF-β1) has a broad spectrum of chondrogenic roles [
2] relevant to cartilage engineering specifically. In native cartilage extracellular matrix (ECM), TGF-β1 is secreted by chondrocytes in its latent form [
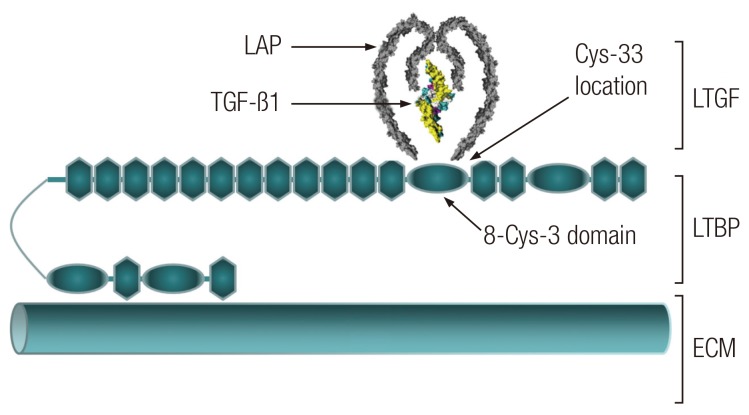
3]. The latent TGF-β1 (LTGF) consists of a mature (active) TGF-β1 associated to its pro-peptide, the latency-associated peptide (LAP), by non-covalent interactions [
4]. LTGF is then bound to the LTGF binding protein (LTBP), itself covalently anchored to the ECM. This complex formation between LTGF and LTBP is mediated by an intra-molecular disulphide exchange between the third 8-cysteine (8-Cys-3) domain of the LTBP and a pair of cysteine-33 (Cys-33) residues of LAP (
Fig. 1) [
5]. The LAP-TGF-β1 association preserves the bioactivity of the growth factor [
6]. The half-life of the LTGF of rats has been reported to be greater than 100 minutes as compared with a short 2 to 3 minutes for the active form [
7]. The activation/release of TGF-β1 involves disruption of the non-covalent interaction between the LAP and TGF-β1 by proteases, thrombospondin-1, low pH and reactive oxygen species [
4,
8]. The LAP also possesses two arginine-glycine-aspartic acid sequences which activate the TGF-β1 on cell attachment [
4]. In general, TGF-β1 stimulates growth in mesenchymal cells and is inhibitory to epithelial and endothelial cells [
8]. The cell response to TGF-β1 varies depending on the dose, length of exposure and target tissue [
8]. A prolonged exposure and high dose of TGF-β1 may result in pathological conditions such as extensive fibrosis, hypertrophic scarring and formation of osteophytes [
8,
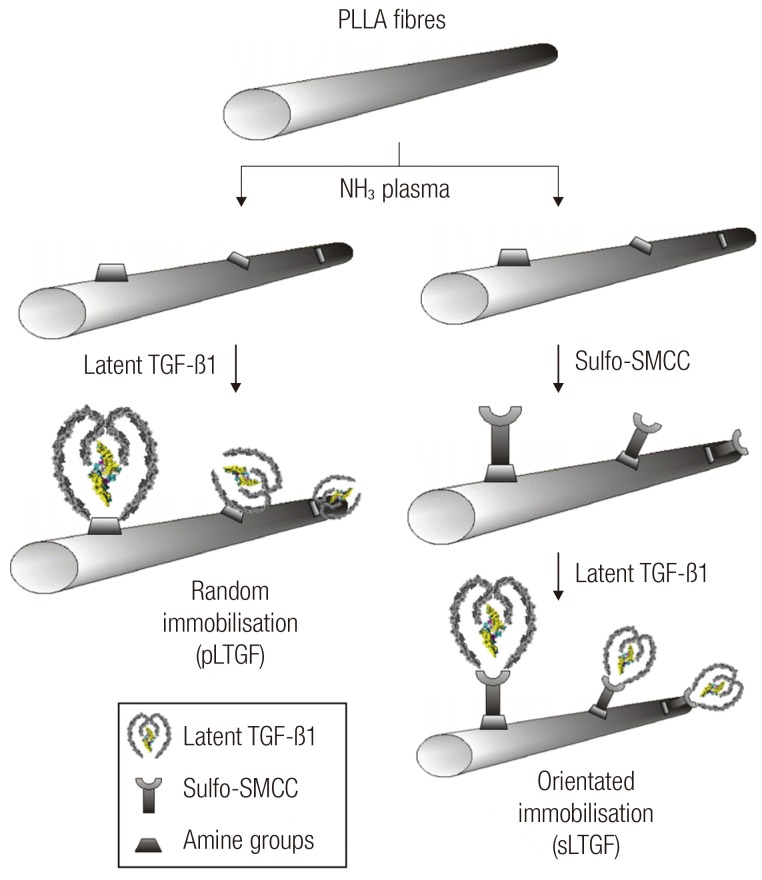
9]. An engineered system capable of inducing cell-mediated TGF-β1 activation will begin to address these problems through cartilage engineering. The novel strategy presented in this study is to incorporate LTGF onto an electrospun scaffold and allow chondrocytes to sense and activate TGF-β1 as required. Two techniques of scaffold biofunctionalisation are described: scaffold surface modification using ammonia plasma, and this combined with subsequent sulfosuccinimidyl-4-(
N-maleimidomethyl)cyclohexane-1-carboxylate (sulfo-SMCC) treatment. The effects of scaffold biofunctionalisation with LTGF on the cell phenotype were evaluated
in vitro and
in vivo using primary human nasal chondrocytes.
DISCUSSION
TGF-β1 has been used extensively to control growth and differentiation in tissue engineering, and a variety of delivery systems have been explored. The concept behind the scaffold system developed in this study is to guide cell-mediated activation. This may allow cells to determine their proliferation and function as necessary, preventing potential TGF-β1 adverse effects. LTGF was incorporated into a novel biomimetic regenerative scaffold relevant to cartilage engineering. Our results confirm that an LTGF biofunctionalised scaffold design successfully influenced chondrocytic behaviour based on Sox9 and Col2A1 expression in vitro and in vivo.
The TGF-β1 immunoassay revealed that the LTGF was successfully anchored onto the plasma treated scaffold surface. The random immobilisation technique was more efficient in anchoring a larger amount of the LTGF (approximately one order of magnitude), as compared to the orientated immobilisation approach. This could be for the following reasons: 1) the LAP has limited free thiol groups; 2) the specific location of the free thiols of the LAP may be sterically hindered; and 3) the short spacer contained in the sulfo-SMCC further limits its scope of reaction with distant thiol groups. The immunoassay also demonstrated that the immobilised LTGF remains available for enzymatic and chemical activation. The bioavailability of the TGF-β1 on the LTGF biofunctionalised scaffolds was further confirmed by primary human chondrocytic culture.
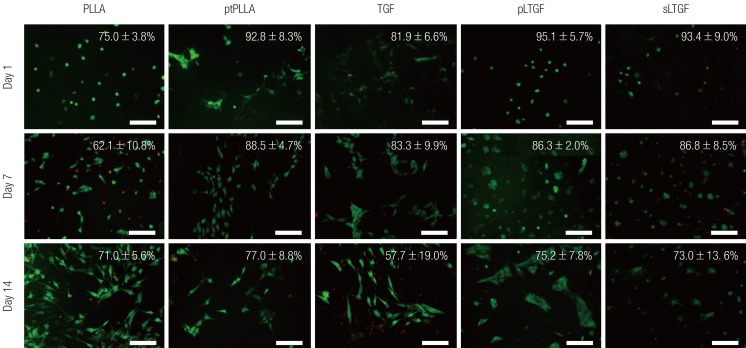
The results of the LIVE/DEAD assay demonstrated that the LTGF biofunctionalised scaffolds are non-cytotoxic and can sustain cell viability of over 70%. The TGF group, despite receiving TGF-β1 (10 ng/mL) supplementation for up to 7 days, failed to sustain the level of cell viability seen in the LTGF biofunctionalised groups. The general declining trend in cell viability over time may be related to the use of SFM. A serum-free environment is used to revert the mitotic cell cycle (proliferation) induced by serum into the interphase cycle (differentiation) [
16]. In native ECM, cells can only express a specific function while they remain in the interphase, and chondrocytes naturally arrest in an infinite interphase cycle [
16]. Once isolated from their natural environment and passaged
in vitro, dedifferentiation occurs-classically characterised by the down-regulation of cartilage-specific genes (e.g., Sox9 and Col2A1) and a gradual rise in Col1A1 (dedifferentiation marker) [
17]. The ability to redifferentiate declines with the passage number. Sox9 is a key transcription factor in chondrogenic differentiation that directly up-regulates the expression of Col2A1 [
18]. Its expression occurs in all chondroprogenitor cells and chondrocytes, except hypertrophic chondrocytes, and has been reported to parallel Col2A1 during embryonic chondrogenesis [
19].
In this study, primary human chondrocytes were cultured up to the third and fourth passages. At such passage numbers, a dedifferentiated phenotype is expected, and Col2A1 was undetectable at 14 days of
in vitro culture. This was consistent with studies which have shown that passaged chondrocytes only re-expressed the Col2A1 gene after 21 days of
in vitro SFM culture [
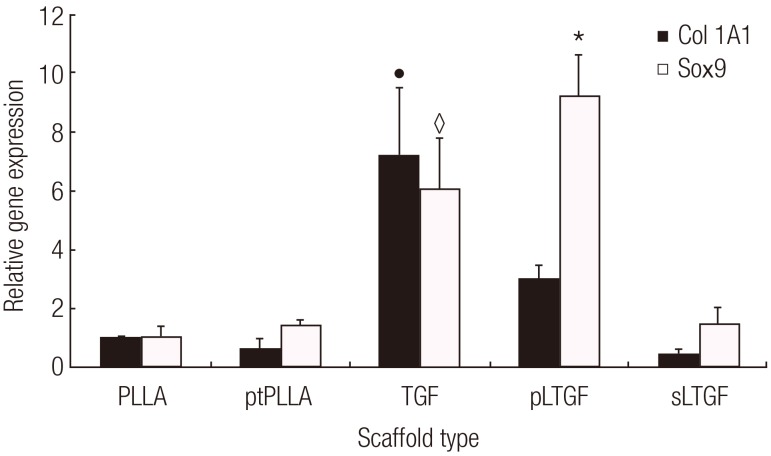
13]. The first indication of redifferentiation at 14 days was demonstrated in the pLTGF and TGF groups with significantly up-regulated cartilage-specific transcription factor Sox9. The up-regulation of Sox9 seen in the TGF group was consistent with a previous study [
20]. Interestingly, the up-regulation of Sox9 in the pLTGF group was significantly higher (approximately 30%) when compared to the TGF group, considering the amount of LTGF available from the pLTGF scaffold (195.4 pg/cm
2 scaffold)-approximately 150-fold lower than the standard TGF-β1 supplementary dose received by the TGF group (10 ng/mL) [
21]. This suggests that although the anchorage of the LTGF onto the pLTGF scaffold may not achieve the intended natural configuration, the LTGF remained protected and available for activation. Although TGF-β1 is known to have positive effects on matrix production and chondrocytic differentiation [
1], some studies have reported its suppressive effects on collagen type II synthesis and the down-regulation of chondrocyte differentiation [
22]. The high Col1A1 expression in the TGF group is consistent with such studies. The sLTGF group did not significantly up-regulate Sox9 gene expression. This may reflect the relatively small concentration of TGF-β1 present in the sLTGF scaffold.
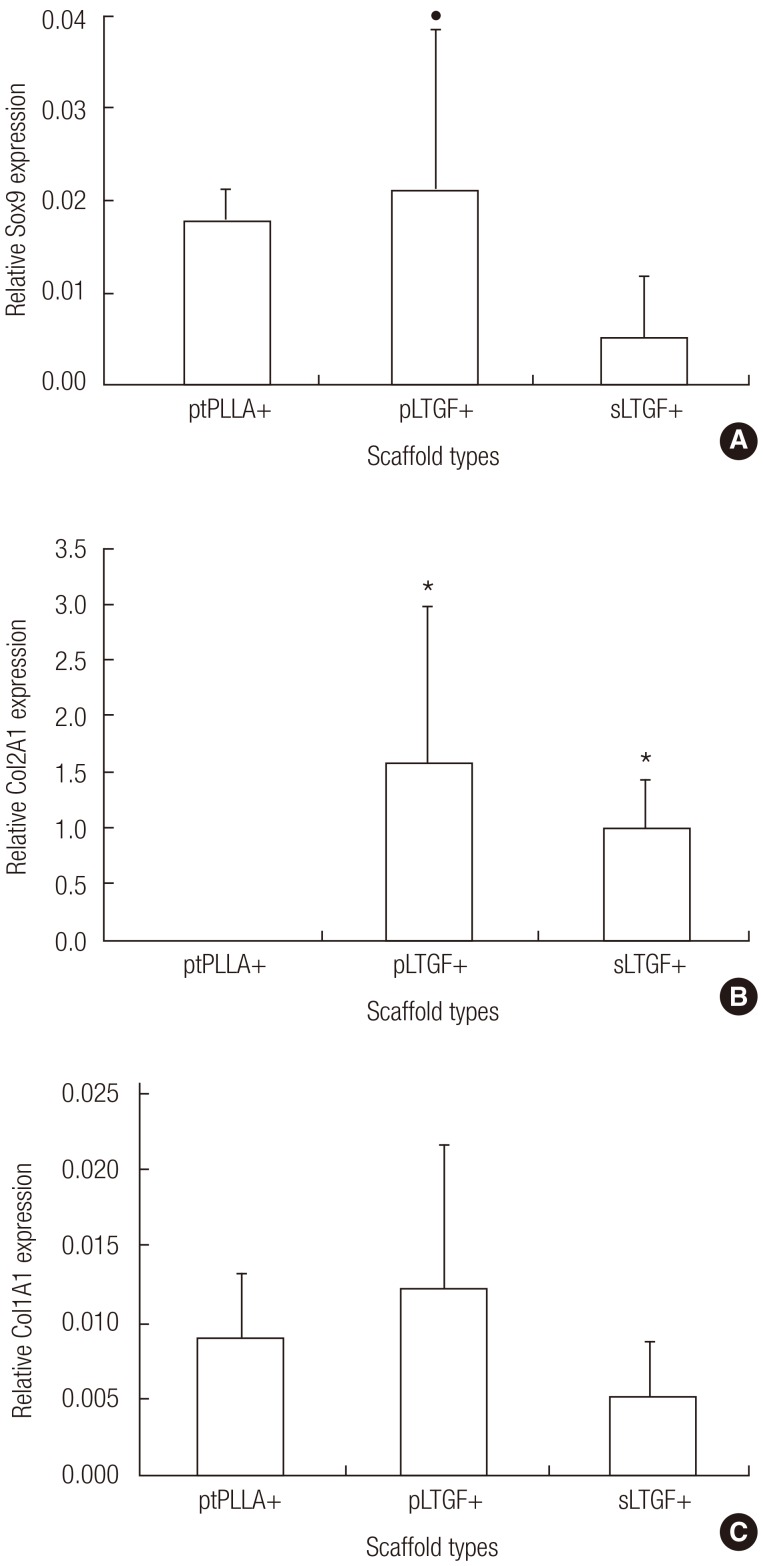
At the end of the 6 weeks of in vivo study, the survival of the donor human cells was confirmed by the detection of human-specific genes by reverse transcription-polymerase chain reaction and anti-human nuclei immunohistochemistry. Of significance, Col2A1 expression was detected on the pLTGF+ and sLTGF+ groups. This confirms that the dedifferentiated chondrocytes are induced to redifferentiate by the LTGF biofunctionalised scaffolds. The expression of Col2A1 was undetectable in the implanted controls (ptPLLA+) despite the presence of Sox9. Although Sox9 is required for the activation of the Col2A1 gene, the results demonstrate that Sox9 expression in the ptPLLA+ group is not sufficient in activating the Col2A1 gene in the absence of the LTGF biofunctionalised scaffolds.
Although Sox9 expression in the sLTGF+ group was significantly lower than in the ptPLLA+ group, Col2A1 was expressed. The level of Sox9 expression seen in the pLTGF+ group, which was 3.9-fold higher than that of sLTGF+, was not accompanied by a proportional rise in Col2A1 expression when compared to the sLTGF+ group. This "anti-parallel" trend between Col2A1 and Sox9 may be a gene profile characteristic of
in vitro passaged adult human chondrocytes, where the expression of Sox9 correlates to a lesser extent with the presence of Col2A1 [
18]. This may be due to a decrease in Sox9 binding activity to the Col2A1 gene enhancer site with increasing passage number [
18].
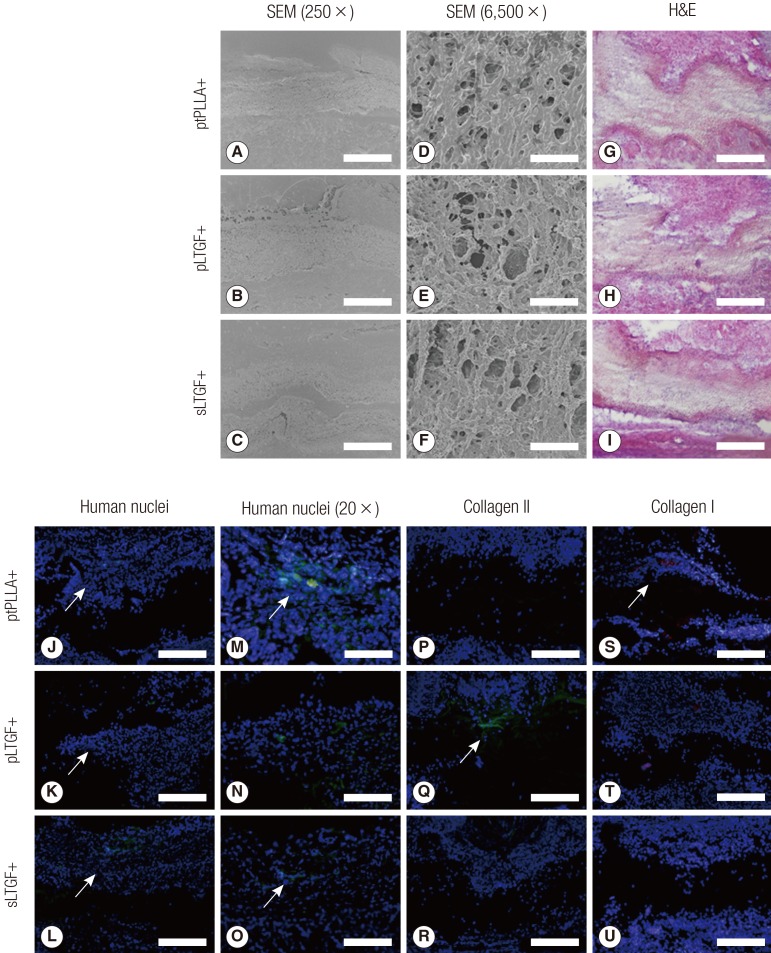
Chondrogenesis in the LTGF biofunctionalised groups was evident in the immunofluorescence detection of type II collagen. Human type II collagen was detected in sections of the pLTGF+ group to further confirm the chondrogenic efficacy of the biofunctionalised scaffolds (
Fig. 9).
Although integration can be seen on the H&E stains and SEM images (
Fig. 9), negative staining for glycosaminoglycan with safranin O (results not shown) might reflect a slow redifferentiation of the dedifferentiated chondrocytes [
23]. In addition, TGF-β1 may reduce glycosaminoglycan synthesis in mature chondrocytes and increase the degradation of aggrecan by up-regulating aggrecanase activity [
24]. This may explain the negative staining of safranin O in all of the scaffold groups. The type I collagen on immunofluorescence, found in the ptPLLA+ group, was consistent with Col1A1 gene expression and lack of Col2A1 expression-and further confirmed that those chondrocytes remained in their de-differentiated state.
We can deduce that in such systems there are two key mechanisms by which TGF-β1 influences chondrocyte behaviour: firstly, by dose, and secondly, by factor presentation (active vs. latent form). A high dose and frequent supplementation of active growth factor may not necessarily result in the desired response (as seen in the low Sox9 and up-regulation of Col1A1 in the TGF group). Conversely, a dose 150-fold lower, coupled with the presentation of the growth factor in its latent form to cells, resulted in a change in the pattern of gene expression of the chondrocytes. Further LTGF dose titration and protein conformational studies need to be performed to verify this argument.
Numerous studies have "immobilised" or "incorporated" growth factors such as TGF-β1 and fibroblast growth factor into tissue scaffold systems with sustained release properties [
25]. These studies have used active forms and inevitably involved processing steps which may have resulted in reduced bioactivity. In addition, the majority of scaffold release systems are hydrogel based, and release of growth factors is by diffusion or degradation of the scaffold matrix. This often results in a "burst" release and a compromise in scaffold mechanical and other properties required for cartilage engineering [
1]. In our strategy, the growth factor remains immobilised on the scaffold and protected by its latent protein until cell activation.
Scaffolds functionalised with a latent form of TGF-β1 have been used here to direct cell-mediated activation of this key growth factor in chondrogenesis. The presence of the growth factor influenced the chondrocyte cell phenotype, and this strategy may well prove useful for other growth factors and more widespread application in the field of tissue engineering.