INTRODUCTION
Autologous fat grafts are widely used for cosmetic purposes, for correcting soft tissue contour defects, and for reconstruction, mainly because of their straightforwardness and relatively few associated problems. The major drawback of this procedure is the high absorption rate of the injected fat, which can be 20% to 70% by volume [
1].
On the fourth day after injection, new angiogenesis from the host vascular network causes graft revascularization [
2], but this process is rather limited to the fat cell mass periphery and centrally located adipocytes have little contact with the vascular supply. Accordingly, the death and removal of the fat cells continue until the reorganized blood supply matches the graft vascular demands, and thus, early and appropriate revascularization is important for graft survival [
3,
4].
Diabetes mellitus is one of the major chronic diseases in nearly every country. The universal prevalence of diabetes among adults (aged 20-79 years) was 6.4% in 2010, and this percentage is expected to increase to 7.7% by 2030 [
5]. Therefore, the number of diabetic candidates requiring an aesthetic fat graft will also gradually increase. However, the circulation complications of diabetes are serious and have been shown to involve microvascular problems, particularly with respect to the impairment of collateral vessel formation [
6]. Furthermore, many experimental studies suggest that diabetes impairs ischemia-driven neovascularization.
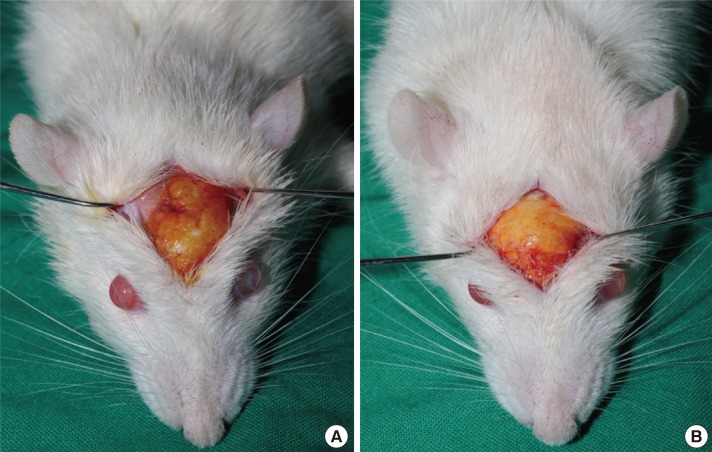
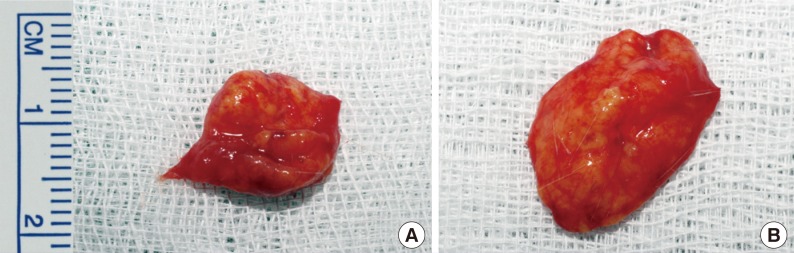
The principal assumption of this study was that the diabetic condition inhibits the survival of the grafted fat by impairing neovascularization. To investigate this assumption, a rat scalp fat graft model was designed to evaluate the fat survival. The effect of the presence of diabetes on the grafted fat was estimated by calculating the volume and the weight of the grafted fat and by a performing histological analysis on the grafted fat sections.
DISCUSSION
Fat injection for reconstruction and cosmetic purposes was first described in the late 1980s [
10], but it was not until the 1990s, when Coleman [
11] outlined a standardized reproducible "process" for fat harvesting, centrifugation, and injection, that the reliability and reproducibility of facial "structural fat grafting" became evident. Subsequently, the use of free autologous fat grafts became immensely popular within the medical field, principally because fat tissue is plentiful, easily harvested, and hypo-immunogenic, and also has favorable physical features [
12], which include biocompatibility, versatility, a natural appearance, and low donor site morbidity. The major drawback of this procedure is the high absorption rate of the injected fat, which is probably the result of ischemia and a lack of neoangiogenesis [
13].
Contradictory theories have been proposed regarding volume maintenance after fat grafting [
14]. The most commonly established of these theories is Peer's cell survival theory [
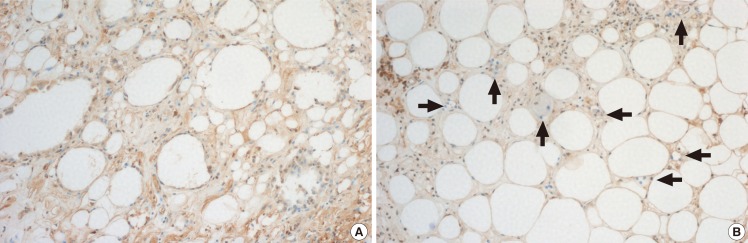
15]. Damage of the grafted fat is processed immediately during and after injection because of the mechanical injures due to the injection force and because devascularization causes ischemic injury. After the fat injection, on the first day, neutrophils infiltrate the graft, followed by macrophages, histiocytes, and multinucleated giant cells. Histologic examinations of free fat autologous grafts indicate minimal fat cell necrosis at 48 hours post-injection, with only partial graft vascularization. On the fourth day post-injection, from the host bed vascular system, neoangiogenesis leads to graft vascularization. However, the process is slightly limited to the peripheral area of the graft, and the central cells can only obtain marginal access. This process of adipocyte death and removal continues until the reorganized blood supply meets the graft vascular demand, and thus, early and appropriate revascularization is crucial for fat survival. Delays in vascular supply to tissues cause adipocyte necrosis, which include cell membrane detachment and the development of fatty cysts and vacuoles in non-vascularized regions [
16]. Thus, vascularization is the most essential factor of graft survival.
Diabetes mellitus is one of the major chronic diseases, and patient numbers continue to increase, as changing lifestyles reduce physical activity and increase obesity. The world prevalence of diabetes among adults between 2010 and 2030 is expected to change as follows: a 20% increase in the prevalence is expected in developed countries and a 69% increase in developing countries. Therefore, the number of diabetic aesthetic fat graft candidates is sure to increase.
However, significant proportions of morbidities and mortalities among the diabetic population are due to a slow and poor response to tissue ischemia [
17]. When chronic tissue ischemia occurs, poor wound healing results in diabetic ulcers and an increase in the lower extremity amputation rate [
18]. On the basis of
in vitro evidence of increased levels of apoptosis in high-glucose cultures [
19] and in cells exposed to hypoxia, it has been assumed that cells exposed to both hyperglycemia and hypoxia show synergistic increases in apoptosis that are expected to increase tissue damage [
20].
In vitro, one study demonstrated that prolonged incubation of diabetic cells in normal glucose cultures did not reverse this phenotype [
17]. In one recent study, diabetic cells were isolated from mice in which diabetes had been induced by streptozocin of 4 weeks duration. This implies that 1 month of diabetes and hyperglycemia can cause cellular changes that cannot be reversed by glucose-level normalization. This also suggests that impaired neovascularization and a broken normal wound-healing mechanism will persist even after diabetes is cured. This glycemic memory has been observed clinically in the context of neovascular damage despite strict prolonged glycemic control [
21]. Thus, we suggest that further studies be undertaken to improve neovascularization and reduce resorption rates to broaden the use of fat grafts in diabetic patients for aesthetic purposes or chronic wound reconstruction.
In terms of experimental human fat grafting, almost every previous study has used the athymic nude mouse model because of its resistance to xenograft rejection [
1,
2,
8,
10,
22,
23]. However, in the present study, we have focused on the hypo-immunogenic characteristics of fat tissue, and processed fat from a human donor in a traditional maneuver and grafted this into Sprague-Dawley rats. Before performing the main experiment, we performed a pilot study on 5 Sprague-Dawley rats per group to observe an immune rejection response. During the 10-week pilot experimental period, no symptoms of hyper-acute or acute immune rejection, such as, infection, redness, edema, or necrosis, was detected. The results of the main experiment concurred with the findings of the pilot study. Nevertheless, the authors are planning further studies to determine the reason for the immune tolerance of the grafted human fat.
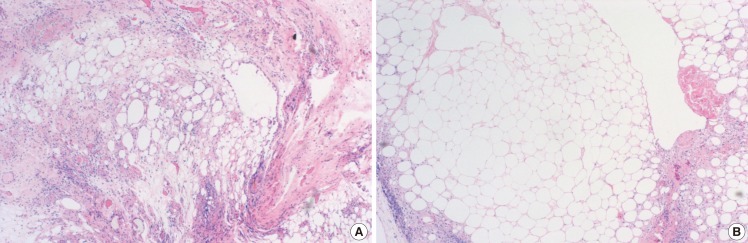
To summarize, the present study shows that the presence of diabetes impairs the survival and quality of fat grafts as evidenced by the lower fat graft weights and volumes and poorer histologic graft qualities in the diabetic group. These findings caution that if a fat graft is unavoidable in a diabetes patient, the possibility of an unfavorable result should be considered. Finally, we recommend that further studies be undertaken to improve neovascularization and reduce the resorption rate to allow the indications of fat grafting for aesthetic purposes or reconstruction to be widened to include diabetic patients.