INTRODUCTION
Breast animation deformity (BAD) is a condition in which the shape of the augmented or reconstructed breast changes or is distorted during contraction of the major pectoralis muscle [
1]. The distortion may lead to varying degrees of widened cleavage, and upward or superolateral implant malposition and asymmetry [
2,
3]. BAD has been reported to occur following submuscular implant placement in breast augmentation procedures and frequently after immediate breast reconstruction [
4-
6].
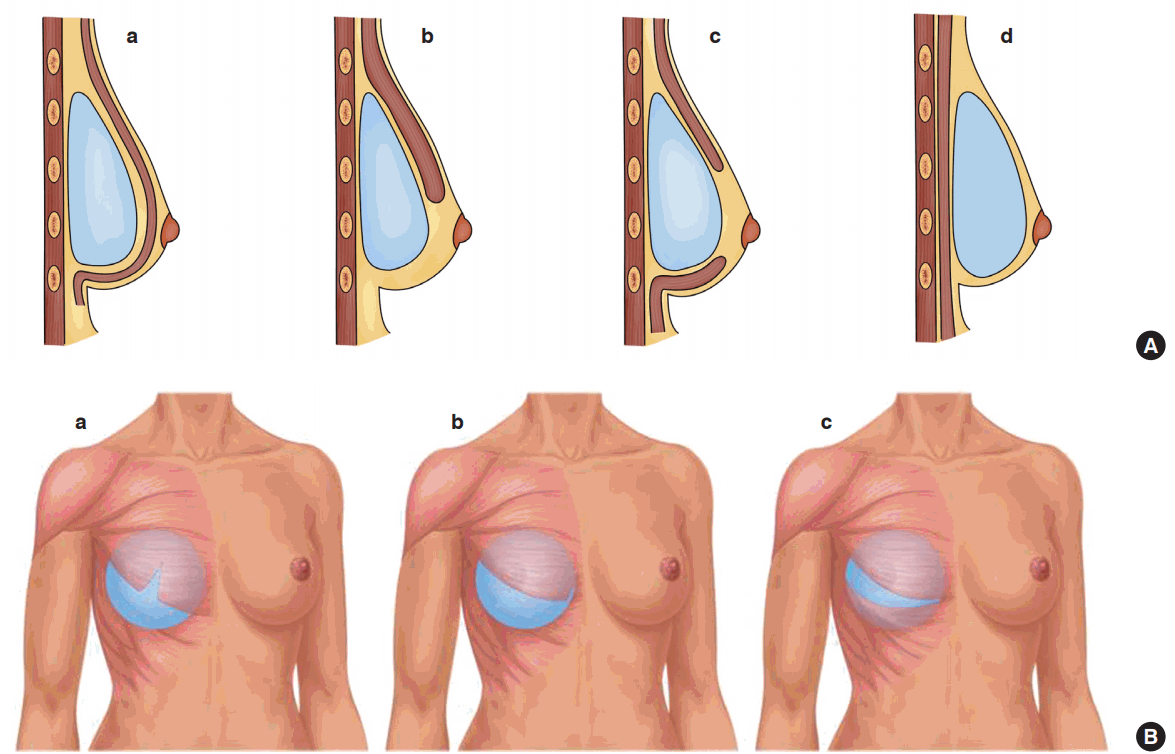
The most commonly employed pocket planes for augmentation mammaplasty are prepectoral (subfascial or subglandular) and partial subpectoral (dual or triple-plane) (
Fig. 1). The opposite tends to apply to breast reconstruction, for which the most commonly employed pocket is subpectoral or submuscular. The reason for this is to place the implants in a space with a vascularized cover due to concerns about post-mastectomy skin breakdown [
7]. The advantages of subpectoral or submuscular placement are reduced rippling and implant visibility, as well as a lower incidence of capsular contracture. Furthermore, it facilitates mammography [
8,
9].
BAD has recently received attention in the literature as a condition that affects especially active women, seems to be significantly more common in patients with subpectorally-placed implants, and appears to be even more common in patients who have undergone breast reconstruction procedures [
10]. Spear et al. [
6] reported that up to 53% of patients experienced some degree of BAD following subpectoral implant placement after dual-plane augmentation, whereas Nigro and Blanchet [
10] reported that 75.6% of patients experienced some degree of BAD after reconstruction. BAD is not only a cosmetic problem, but also a functional problem, as it can lead to muscle twitching, pain, and possibly impaired shoulder function in physically active women, which may ultimately affect their quality of life [
2,
5]. Furthermore, BAD may be associated with a longer postoperative recovery time, as more pain has been reported in patients who underwent subpectoral reconstruction and augmentation than in their counterparts who underwent prepectoral reconstruction and augmentation [
11,
12].
Various methods of treating BAD have been investigated, including neuromodulating injections into the pectoralis major muscle [
13], selective nerve transection [
14], reoperation with a muscle-splitting technique [
15], and repositioning of the implant to a prepectoral position with or without fat grafting [
16].
Prepectoral implant placement is gaining wider acceptance worldwide; however, this implies less tissue coverage, leaving the implant more visible, especially following immediate breast reconstruction and to a lesser degree following breast augmentation. Furthermore, the incidence of capsular contracture has been reported to be higher following prepectoral implant placement [
17]. The use of biological meshes has been described as a way to reduce implant visibility and BAD; however, these materials cannot be expected to replace either the muscle volume or coverage of the implant [
11].
Numerous papers have mentioned BAD and concluded that it is not a significant problem. However, it is much rarer for papers to define BAD, to classify it, or to describe how the degree of BAD was assessed [
3,
14,
16,
18].
The primary aim of this systematic review was to identify and select papers that clearly defined and classified BAD and described how the degree of animation was assessed. Additionally, by utilizing these selected papers, this review aimed to estimate the incidence of this condition following breast augmentation and breast reconstruction. The third aim was to synthesize existing knowledge about BAD and its current classification.
DISCUSSION
Four different surgical methods and types of pectoralis major muscle involvement were presented and compared: (1) the Regnault technique; (2) dual-plane partial muscle coverage; (3) the dual-plane technique in combination with muscle-splitting and partial muscle coverage; and (4) the triple-plane technique with partial muscle coverage (
Fig. 1). The percentage of patients with BAD varied between 30% and 78%, with a median of 58%. Upon first impression, the data seem to indicate that there was no meaningful difference in the incidence of BAD between the Regnault technique and the dual-plane technique, whereas the degree of BAD was lower following the muscle-splitting and triple-plane techniques. When examining the degree of severe BAD, there was a clear trend for the incidence of severe BAD to increase with the degree of muscle coverage, as the use of the Regnault technique by Pelle-Ceravolo et al. resulted in an incidence of severe BAD of 47%. In contrast, Nigro and Blanchet [
10] and Spear et al. [
6] reported that severe BAD occurred in 5% and 11% of patients following dual-plane augmentation and reconstruction, respectively; Pelle-Ceravolo et al. reported that severe BAD occurred in 5% of patients in whom the dual-plane muscle-splitting technique was used; and Bracaglia et al. reported severe BAD in 0% of their patients in whom the triple-plane technique was used. We postulate that the likelihood of BAD varies with the degree of muscle coverage/involvement and that the incidence of BAD can be expected to be close to 0% when there is no muscle coverage. However, this conclusion cannot be firmly made on the basis of this study due to the lack of data of sufficient quality for comparison, and this issue remains to be elucidated.
Thus, the data seem to show a clear association between the degree of muscle coverage and the degree of BAD, even though the grading systems are not directly comparable. Pelle-Ceravolo et al. used a 3-point scale to evaluate their own technique in comparison to the Regnault technique, whereas the three other studies used 4-point scales for their evaluation. This could imply that the incidence of moderate to severe BAD was overestimated in the study by Pelle-Ceravolo et al., since a larger proportion of their patients could have been classified in the severe category. Furthermore, the ability to compare the surgical technique developed by Pelle-Ceravolo et al. with the Regnault technique is limited, as the larger number of patients who underwent surgery using their technique (302 patients vs. 46 patients) might have lowered the incidence of severe BAD.
The examinations of BAD should be compared with caution. The degree of BAD was not evaluated using the same scale, and the evaluation was performed in different ways. Pelle-Ceravolo et al. performed a physical exam for all their patients, whereas Spear et al. and Bracaglia et al. used photographs to evaluate the degree of BAD. The classification of BAD differed among studies. Pelle-Ceravolo et al. used three examiners for each patient: a plastic surgeon, a nurse, and the patient. Spear et al. used a group of plastic surgery residents, and Bracaglia et al. used three experienced plastic surgeons. This combination of different observers, with different levels of experience and expertise, could affect the quality and comparability of the evaluations.
In two of the studies, by Nigro and Blanchet [
10] and Spear et al. [
6], the degree of BAD was evaluated by the patients using questionnaires. Spear et al. reported a remarkable similarity in the assessments of BAD using the photo evaluations and the questionnaires.
The patient questionnaires used in the study by Spear et al. are interesting in that none of the patients experienced any inconvenience in relation to daily activities, but 24% and 19% stated that they were affected when lifting weights or exercising in general. When asked if they would choose the retropectoral implant placement again, 70% would, 28% were unsure, and 3% would not choose it again. However, we cannot know if these data are representative of the entire cohort, since the response rate to the questionnaire was rather low (35%; 69 of 195). This implies a major risk of selection bias; in particular, it is possible that the patients who felt most positively about their outcomes might have replied, while those who were less satisfied did not.
In the most recent study by Nigro and Blanchet [
10], 84 of 108 patients (78%) replied to questionnaires regarding a self-assessment of the degree of BAD and how bothersome BAD was to their daily activities. Three-quarters (74%) of patients reported experiencing some degree of BAD. They did not find a significant difference in the degree of BAD between athletic and non-athletic women. This finding is counterintuitive, as one would expect that BAD would affect athletic women the most, which has also been suggested in two studies that found that athletic women were more prone to suffer from BAD [
6,
14].
The follow-up differed among studies, ranging from 6 months to 15 years. This may have affected the results, as we do not know whether BAD improves or worsens with time.
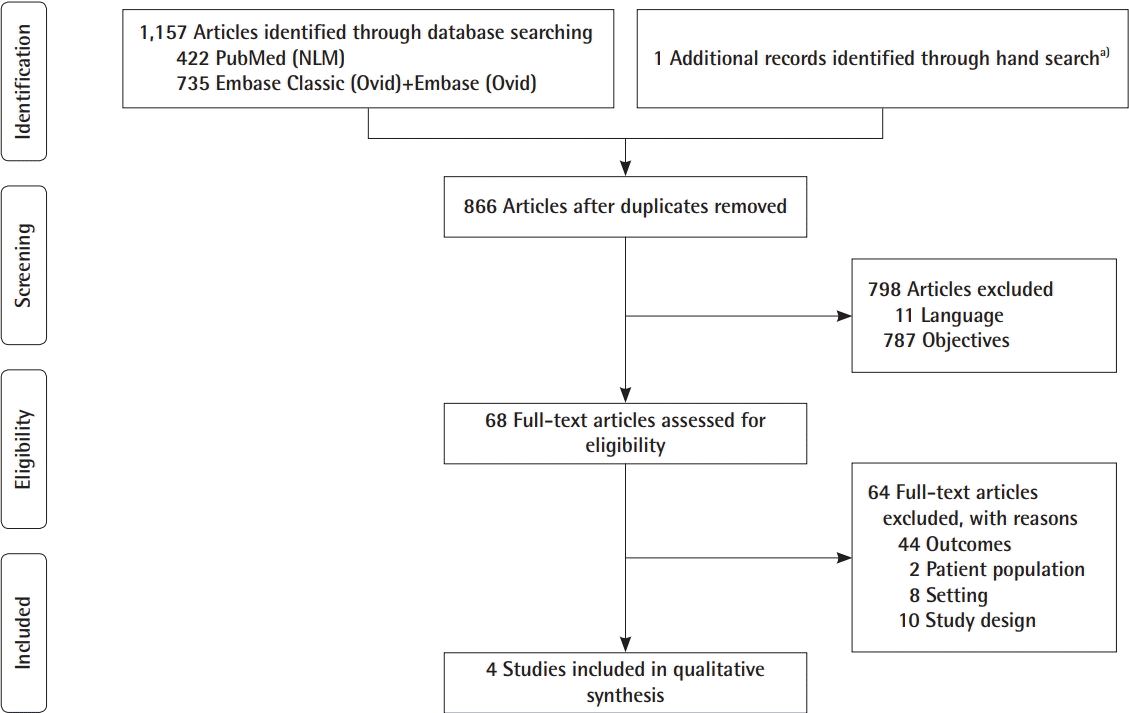
We expected to be able to compare the above-mentioned techniques for implant-based breast augmentation and breast reconstruction to prepectoral implant placement, since the number of papers describing prepectoral implant placement to avoid BAD are increasing at a rapid pace. Unfortunately, several studies were not included in this review because BAD was not clearly defined or described, or because the Results section did not contain any data regarding the assessment of BAD [
2,
3,
5,
11,
14-
16,
18,
21-
27,
29,
30]. The selection process is presented in the PRISMA flowchart in
Fig. 1.
It is challenging to compare the four included studies and to interpret the reported incidence rates due to differences in the assessment of BAD and because the authors did not examine whether their results were reproducible by conducting an inter- or intra-rater reliability study.
The most important question regarding this issue is whether patients perceive BAD as a problem and how it affects their quality of life. A recent study by Becker and Fregosi [
5] investigated this question. They contacted 25 women who had undergone submuscular breast reconstruction and found that 100% experienced BAD and 80% were bothered by it. Although the study population was small, it is noteworthy that all the patients experienced BAD.
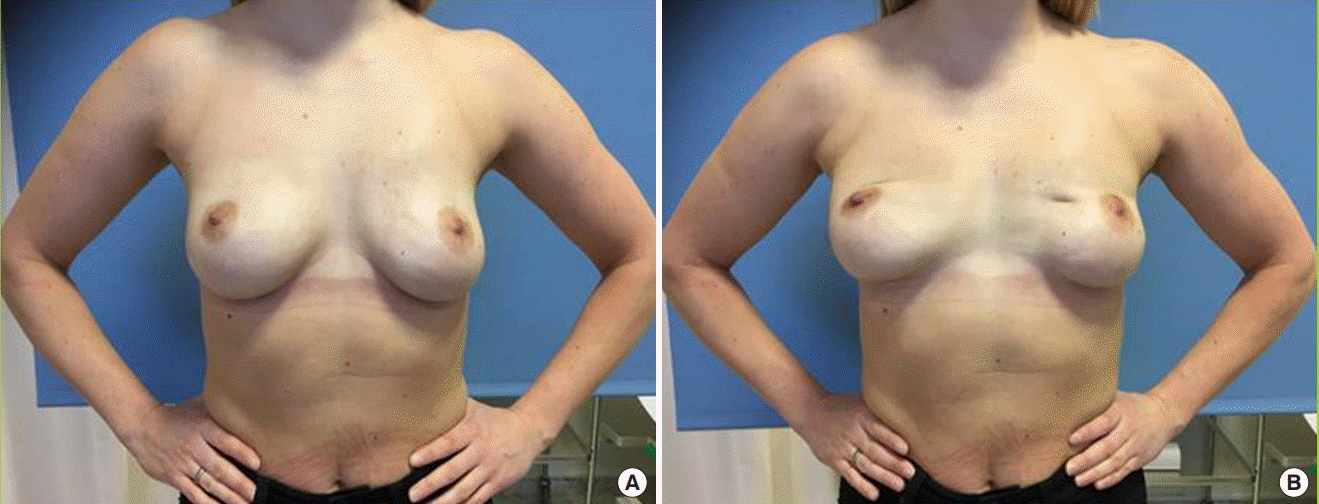
To evaluate this condition, it is imperative that assessments are consistently performed in a reproducible manner to enable a standardized comparison of the results. We need a standardized grading scale for BAD that could be used to assess the degree of BAD in both augmented and reconstructed populations. The optimal assessment would be based on a clinical evaluation in an outpatient clinic. The second-best option is to assess BAD by watching videos of the patients. This may be advantageous, since the videos can be used for inter- and intra-rater reliability assessments to test the reproducibility of the scale. Still photos can be used as a tool for documenting the dynamic phenomenon of BAD if images are taken with the patient in a relaxed position and when activating the major pectoral muscle for comparison (
Fig. 3).
Furthermore, it is necessary to demonstrate that the trend towards prepectoral implant placement, especially in breast reconstruction, can lead to the same level of aesthetic and functional results as submuscular implant placement in the long term. Prepectoral placement of implants might improve BAD, but it may cause significantly more capsular contracture [
17]. However, we will have to wait for at least 5 years before we will get any indication of whether this is true.
Assessment of quality of life and patient-related outcome measures in patients diagnosed with BAD is just as important as grading the severity of BAD itself. We may not need to treat BAD or to change our reconstructive techniques if BAD does not influence quality of life and patient-related outcome measures. We need more studies that assess BAD using quality of life and patient-related outcome measures, as well as a standardized and reproducible grading scale, to gain insight into these issues.