INTRODUCTION
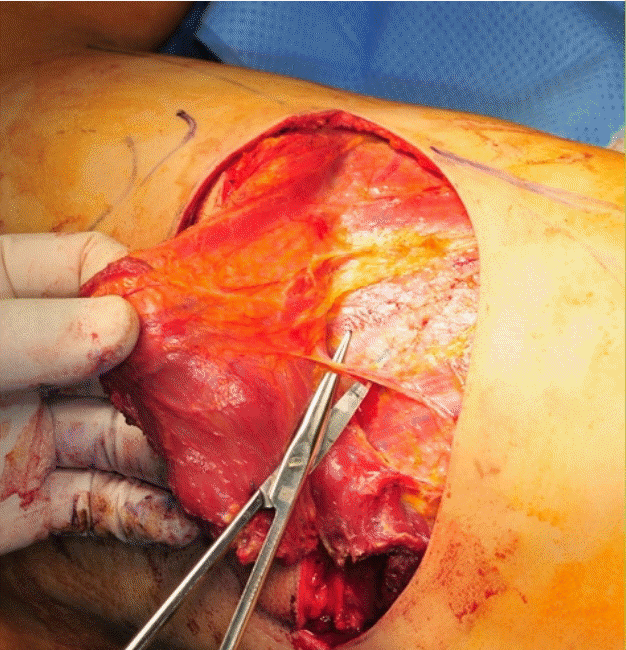
Some vessels with a large diameter are easily found when raising a latissimus dorsi (LD) flap for procedures such as breast reconstructive surgery (
Fig. 1). These vessels include the dorsolateral branch of the posterior intercostal artery (DLBPI). The DLBPI appears to be large enough to be used as a pedicle of a free flap, which is a compelling possibility because doing so would completely conceal the donor scar under the bra band, unlike the thoracodorsal artery perforator (T-DAP). This would be advantageous for patients’ quality of life, but no report has yet described the use of this branch as the pedicle of a free flap.
The location of the scar at the donor site after breast reconstruction is a critical issue for women. Thus, reports have described flap harvesting designs that can hide the donor site underneath the bra band [
1-
3]. The authors of the present study employ such designs for breast reconstruction and have observed that patients tend to exhibit interest in the location of the eventual scar tissue.
Pedicled island flaps using the dorsal branch of the posterior intercostal artery were described in a study that analyzed intercostal artery perforator flaps [
4]. However, the dorsolateral branch was not clearly distinguished from the dorsal branch of the posterior intercostal artery in that study. Subsequent anatomical research has yielded more information about the DLBPI, including its location and origin [
5]. However, to our knowledge, it remains unknown whether this branch can be used as the pedicle of a free flap and whether this branch alone can provide perfusion to the regional skin.
The purpose of this study was to clarify the anatomy of the DLBPI through a cadaveric study and to assess the usability of the flap through the results obtained when applying it in clinical settings.
DISCUSSION
In 1987, Taylor [
7] reported that detecting angiosomes plays a pivotal role in identifying perforator flaps. They also demonstrated that vessels supplying the skin can be used to create a flap. Angiographic studies have identified angiosomes and proven that various body parts can be used as donor sites for free skin transfer, such as in the form of a perforator free flap [
8]. Pedicled perforator flaps have been widely used, and perforator flaps are used for free skin transfer.
To use a free flap with the DLBPI, the authors thought that an anatomical study would be necessary and accordingly used 10 cadavers to check the location and distribution of the DLBPI. Offman et al. [
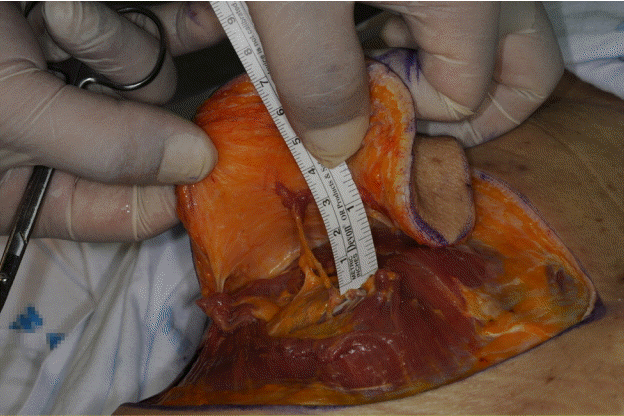
9] reported that the perforator from the posterior intercostal artery was variable, but in this study, all perforators could be observed from the seventh to eleventh intercostal spaces. Thus, it was possible to find a pedicle measuring 9.18 cm on average from the scapular tip, 9.79 cm on average from the spinal process, and 4.82 cm on average laterally. Despite differences between Western and Asian populations, this finding was similar to the result reported by Prasad et al. [
5] that the perforator had a pedicle measuring an average of 4.6 cm in the eight to eleventh intercostal spaces. In our study, dissection of the cadavers revealed that the average number of DLBPIs (diameter ≥2 mm) was 1.65±0.67, and that they were located between the 7th and 11th intercostal spaces. The DLBPI was easily found in all cadavers dissected in the study and was reliably located in the specified area.
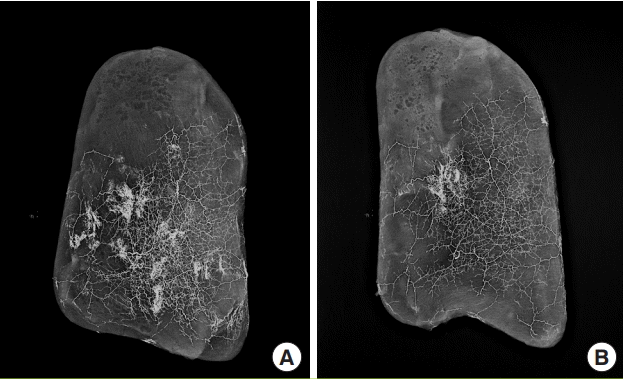
In the cadaveric study, we investigated the extent to which the dye spread using mammography, which involves a greater dose of radiation than ordinary X-ray scans. Our initial speculation was that when the dye was injected into the DLBPI, it would spread throughout a limited area, which would show the perfusion capability of the branch. Concerns were raised that the injection pressure, the viscosity of the dye, and the concentration of the dye could act as confounding factors affecting the extent to which the dye would spread. However, when the injection was made, the dye diffused throughout the system easily. Immediately after the injection, the dye spread throughout the entire system, overflowing the margins. Based on this observation, the vessel network around this area appears to be very well developed. Nonetheless, perfusion to the skin layer is presumed to be supplied through the muscle layer for the following reasons: the dye spread throughout the area corresponding to the LD muscle both when the MC flap was present and when only the skin layer was imaged, and countless small vessels between the skin and muscle were observed when the LD muscle was removed from the skin layer.
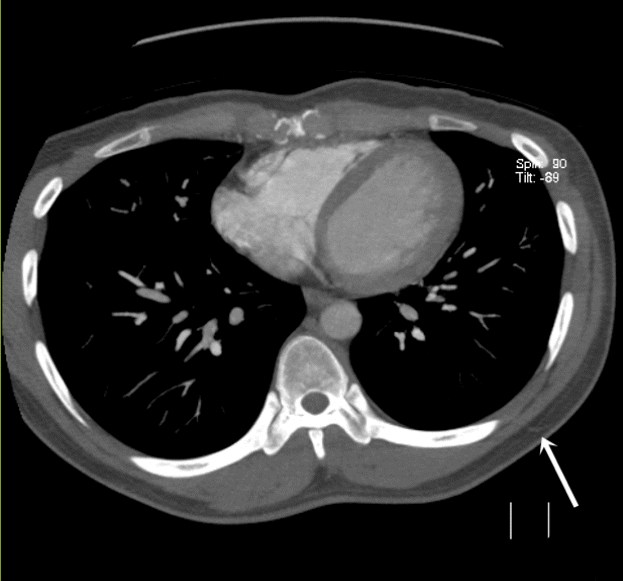
Based on these anatomical results, it was concluded that the location of the perforator could be found using Doppler ultrasonography preoperatively, with the patient in the sitting position, and the perforator was confirmed through chest CT (1-mm fine sections). Therefore, it was deemed possible to design the DLBPI flap using a pinch test centered on the location of the perforator determined by Doppler ultrasonography. Subsequently, reconstruction with this flap was conducted in patients with defects, who were considered suitable for anastomosis with a short pedicle in various areas.
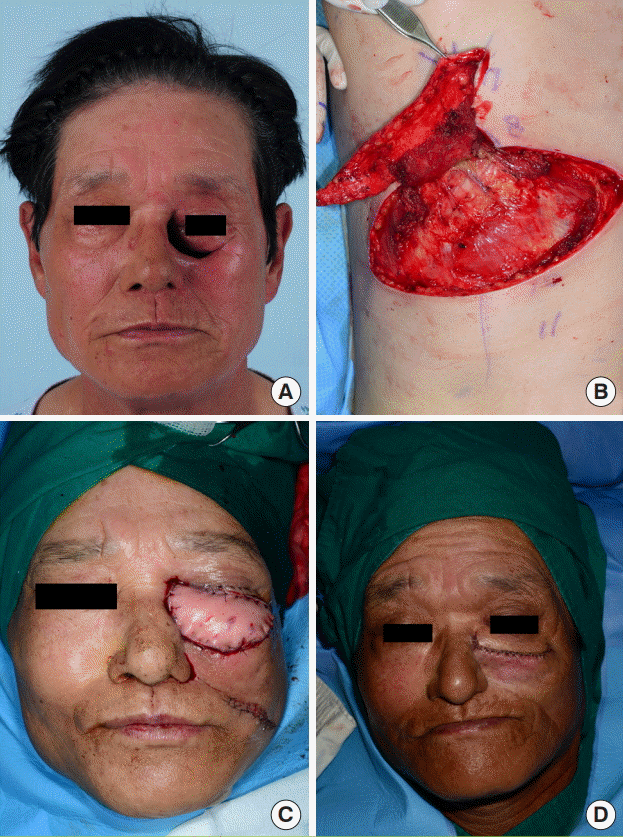
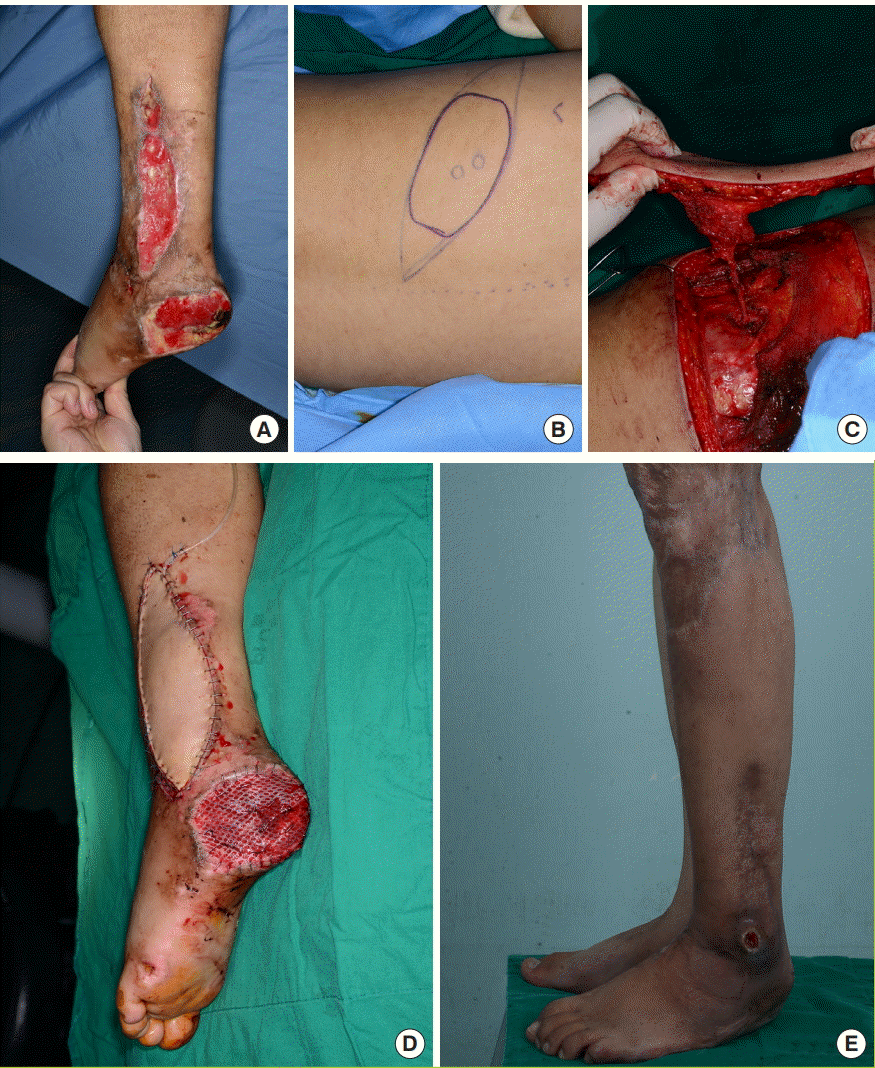
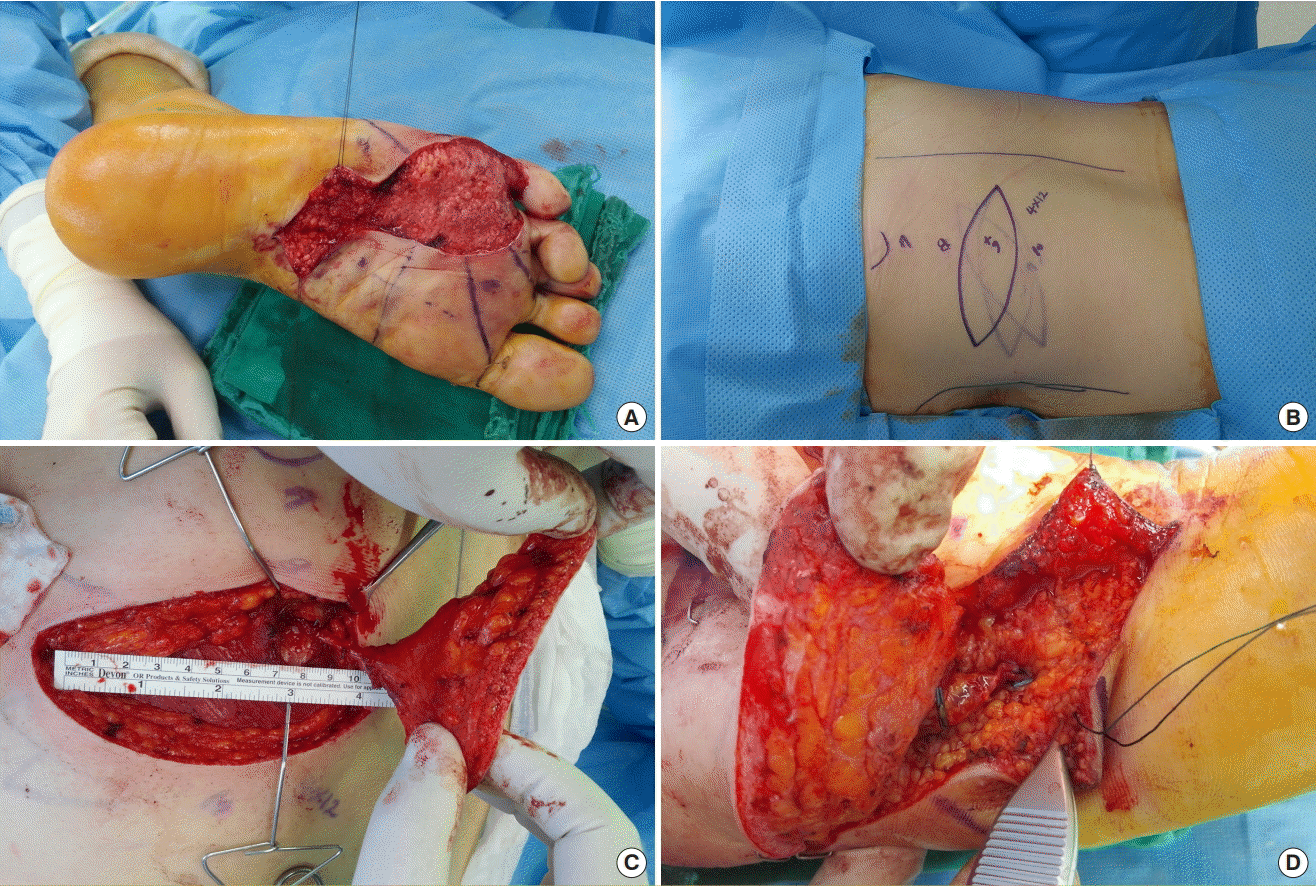
Several considerations must be taken into account when performing a free flap with the DLBPI in clinical settings. First, the pedicle of the DLBPI free flap is short (4–5 cm). Therefore, the recipient vessel must be located close to the defect. Moreover, if the condition of the recipient vessel at the time of anastomosis is not optimal, the procedure cannot be performed successfully. The DLBPI flap for lower extremity reconstruction involved perforator-to-perforator anastomosis, but anastomotic failure occurred due to a spasm of the recipient’s vein (case 2) or artery (case 3). In case 2, the flap showed total necrosis at 10 days postoperatively, after which a secondary operation with a skin graft was required. In case 3, the recipient arterial spasm was not corrected in 10 hours despite several efforts. Thus, we had to use the DLBPI flap as a full-thickness skin graft. Second, in patients with a thick subcutaneous fat layer, the flap was too thick, making it burdensome to use this flap (case 1). Third, it has been reported that elevating the flap by dissecting the posterior intercostal artery medially or laterally along the lower costal margin allowed the pedicle to be elongated to as long as 12 cm [
5]. However, the intercostal muscle would be injured in such a procedure, which would become excessively complex, and the pedicle would be likely to be damaged (as the pedicle has many branches in the muscle layer), potentially causing adverse events such as spasms. Therefore, for extension, it is considered much easier to use another free flap (e.g., an anterolateral thigh flap) (case 1). Fourth, the DLBPI may be branched when it inserts into the LD muscle. Such cases require meticulous care, and further dissection slightly deep to the origin in the intercostal muscle can be helpful. Fifth, the DLBPI free flap is safer for use as an MC flap. As previously discussed, it is speculated to be perfused via the LD muscle layer. Therefore, unlike the T-DAP flap or the anterolateral thigh flap, this flap is risky if much of the muscle layer is removed to skeletonize the pedicle or in an attempt to make the flap thinner. Finally, the patient must be placed in the lateral decubitus position during surgery for flap harvesting.
This study investigated the possible use of the DLBPI as the perforator of a free flap. Despite its limitations, the DLBPI free flap may be considered as an alternative free flap for small defects. The major advantage of the DLBPI free flap is that harvesting it is easy and simple, because the DLBPI can be easily found in a reliable location, with rare anatomical variants. Additionally, for women, it has the advantage of concealing the donor scar underneath the bra band, which is beneficial for patients’ quality of life.