INTRODUCTION
Connecting not only the vascular supply, but also sensitive nerves, represents an appealing option in free tissue transfer [
1]. However, in certain cases, such as breast or extremity reconstruction, the feasibility of coaptation of sensory nerves of the flaps remains questionable [
1].
Although the coaptation of sensory nerves in soft-tissue reconstruction with free flaps is frequently conducted [
2-
6], spontaneous return of sensation without sensory nerve coaptation has been reported [
7]. Non-sensate free muscle flaps (e.g., the gracilis muscle flap, latissimus dorsi muscle flap, or rectus abdominis muscle flap [
8]) with free skin grafts represent an attractive alternative, and are common procedures in reconstructive microsurgery [
9]. Excellent rates of spontaneous reinnervation in skin grafts over free muscle flaps that have been sensory-neurotized after sensory nerve-to-motor nerve transfer were recently found in both human and animal studies [
10]. Restoration of the sensitivity of skin that has been grafted onto the transferred muscle is of vital importance [
9], but it has been postulated that free muscle flaps are fully denervated tissue, and that satisfactory innervation of the overlying skin graft cannot be expected. Reinnervation occurs mostly through the surrounding tissue if no nerve branch is sensitized, and it may take a long time to develop. Therefore, functional sensation is hardly restored, nor is adequate tactile sensation or two-point discrimination (2-PD) achieved [
11].
The aim of this study was to evaluate sensitivity in skin grafts over free, non-neurotized gracilis muscle flaps for lower extremity reconstruction.
RESULTS
The indications for the free gracilis flap, defect size, localization, co-morbidities, and outcomes are summarized in
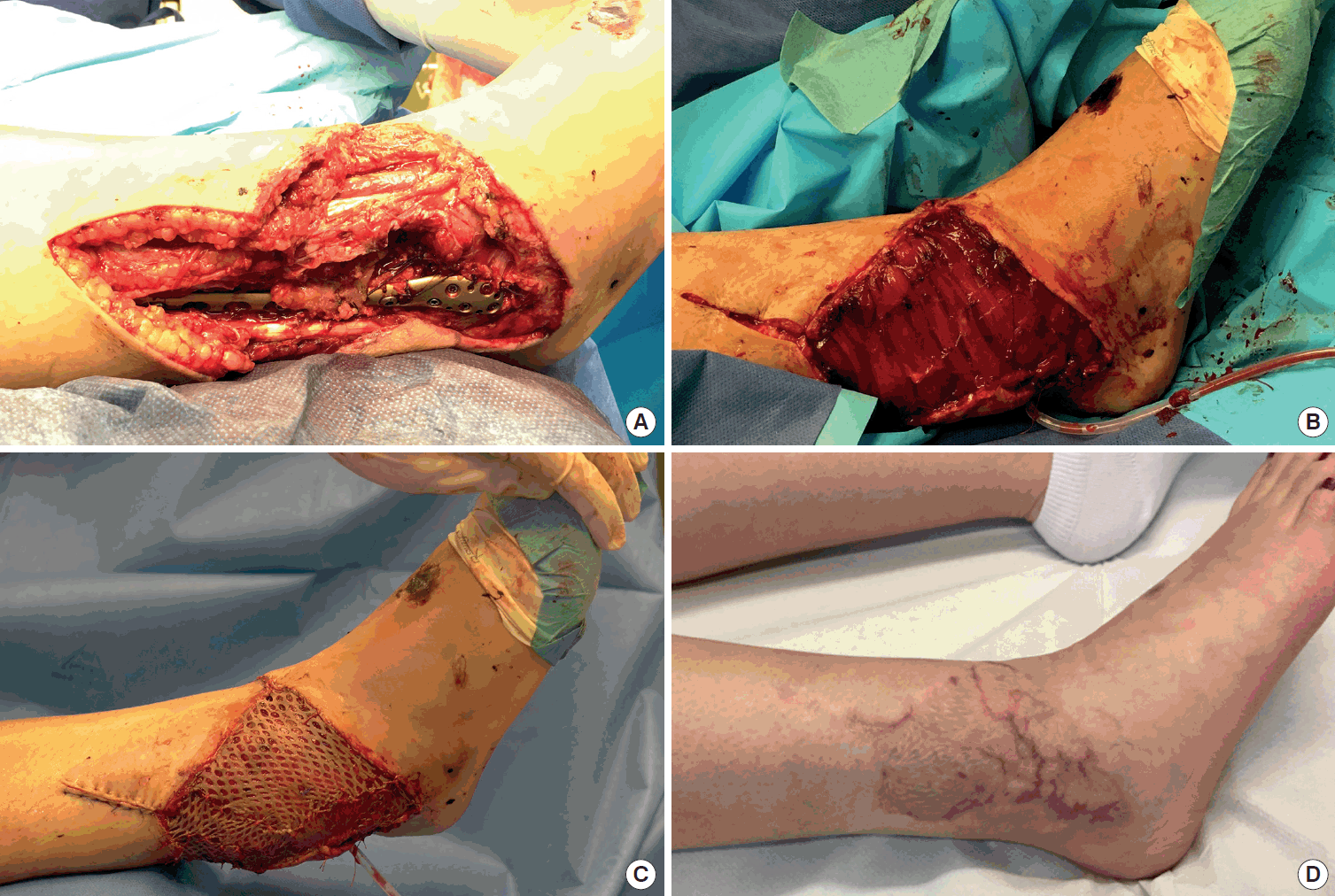
Table 1. In all cases, the volume of the muscle flaps (median flap weight, 114 g; range, 32–225 g) was sufficient to cover the defects (
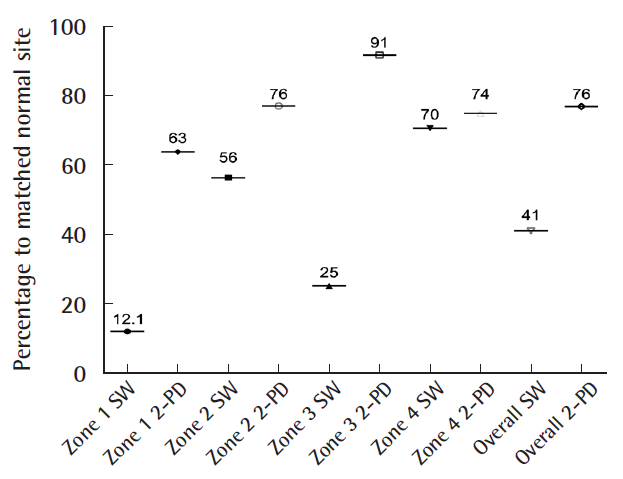
Fig. 2). In six patients, the gracilis muscle flap was trimmed accurately to fit precisely into the recipient site (median resected muscle weight, 33 g; range, 5–90 g). All flaps survived completely without any marginal necrosis or hematoma, and the skin graft take was 90%–100% within 2 weeks in all cases (no grafts were lost to infection). In one patient, wound dehiscence and infection that required revision occurred 1 month after surgery. Three patients presented with ulcerations or hyperkeratosis over the ankle at the final follow-up. Satisfactory contours were achieved for both the patient and the surgeon (11 of 13 patients and 10 of 13 patients, respectively). Seven patients indicated that they were not restricted in the choice of footwear. After a median follow-up time of 14 months (range, 10–51 months), eight of the 13 patients reported normal skin perception. Overall, the SW monofilament test and static 2-PD showed intermediate recovery compared with the corresponding healthy site (41% and 76%, respectively) (
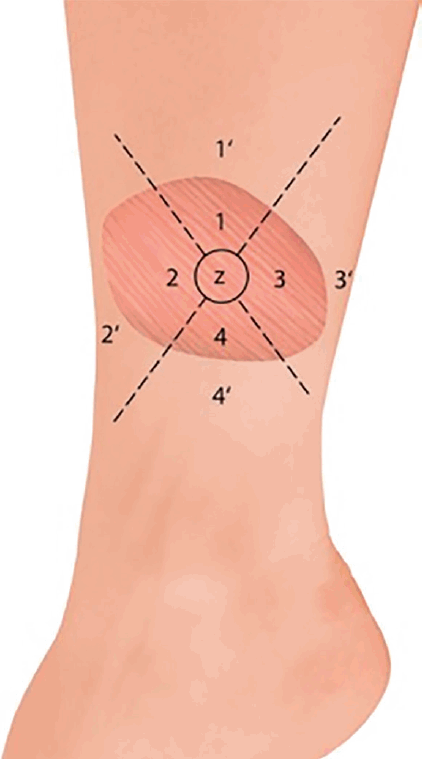
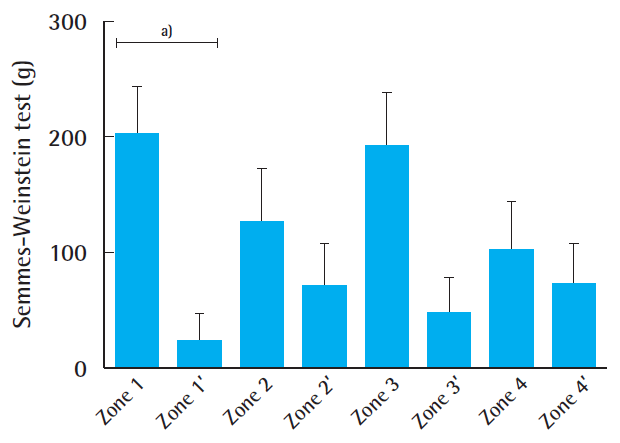
Fig. 3). The SW monofilament measurements only showed a significant difference compared with the corresponding healthy site in zone 1 (201±42 g vs. 24.24±23 g, P<0.05) (
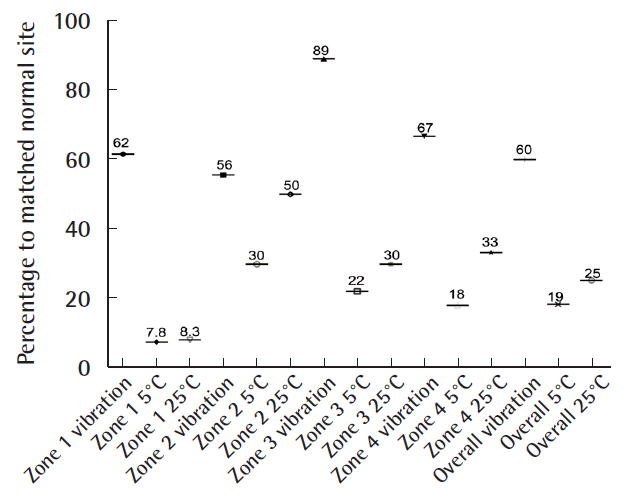
Fig. 4). The highest values were found for the middle of the flap (zone Z, 300 g±0). The 2-PD measurements revealed no significant differences between the flap zones and the corresponding sites, with the lowest pressure sensation in the middle. Five patients had previous surgery on the affected limb, but they showed similar sensory recovery compared with the remaining patients (14.7 mm vs. 13.4 mm for 2-PD measurements and 201.3 g vs. 157 g for the SW monofilament test, similar vibration and thermal sensation). We found an intermediate correlation between flap size and sensate recovery (r=0.27, P=0.36 for the mean 2-PD measurements; r=0.45, P=0.12 for the mean SW monofilament measurements). While overall vibration sensation was restored to a similar extent (60% compared with the surrounding tissue), overall thermal sensation remained poor (19% at 5°C and 25% at 25°C, respectively) (
Fig. 5). In the middle of the flap (zone Z), only three patients were able to feel vibration and thermal sensation (two patients felt 5°C and one patient felt 25°C).
DISCUSSION
One of the ultimate goals of lower extremity reconstruction is a cosmetically pleasing contour, with preserved sensation where possible [
1]. The method of reconstruction should be chosen according to the local requirements and the location of the defect [
4]. Different algorithms have been suggested for the selection of the ideal flap for lower extremity defect reconstruction. In elderly patients, nerve regeneration after nerve coaptation is limited due to small vessel disease, age, or polyneuropathy in patients with diabetes, for example [
1]. Therefore, potentially, young patients can receive free fasciocutaneous perforator flaps or muscle flaps for three-dimensional defect reconstruction, sensate if possible, whereas elderly patients benefit more from non-sensate gracilis muscle flaps [
1].
Nevertheless, we noticed hyperkeratosis and ulceration in three patients over the ankle, which might be a potential drawback of free muscle flaps with skin grafts over friction zones [
15]. The lack of sufficient superficial cutaneous sensation might be a significant contributor to ulceration, even in cases of deep pressure sensation [
15]. Eventually, skin grafted muscular free flaps over friction zones may need to be replaced with sensate fasciocutaneous flaps [
15]. A further potential limitation of muscle flaps might be long-term soft-tissue instability, especially in weight-bearing areas, compared to fasciocutaneous flaps [
16,
17].
It has been reported that free muscle flaps with sensory or motor coaptation can regain sensitivity [
18]. To date, the possibility of sensory restoration of grafted skin has been proven in animal models on a trimmed gracilis muscle surface after sensory nerve-to-motor nerve transfer [
9]. Moreover, Gordon et al. [
19] and Chang et al. [
18] attempted to achieve sensory restoration in a muscle flap with skin grafts using a local sensory nerve that was coapted to the motor nerve for heel and foot sole restoration; however, assessing the critical role of the sensory reinnervated skin graft on a muscle flap proved difficult.
Our results indicate that spontaneous reinnervation of skin grafts on free, non-neurotized muscle flaps is possible. Furthermore, these findings contradict the pre-existing dogma that free non-neurotized muscle flaps are denervated tissue. We observed better recovery of sensory innervation in the peripheral region of skin grafts, confirming the regeneration pattern from the skin margins of the flap to its center. However, although 60% of vibration sensation was recovered (as compared with the surrounding tissue), thermal sensation recovery remained poor. Potentially, vibration may also be perceived through the surrounding tissue, especially in smaller flaps. Nevertheless, our findings might extend the existing principles of lower extremity reconstruction, especially in patients requiring minimal donor site morbidity, a hidden donor site scar, and solid anchorage without shearing forces [
12].
Although our results indicate spontaneous and functional sensation of the skin graft on free, non-neurotized muscle flaps, the results are still inferior to those of sensate fasciocutaneous flaps, but apparently superior to those of non-sensate fasciocutaneous flaps. Still, a precise comparison with corresponding results for fasciocutaneous flaps in the existing literature is difficult, considering differences in procedures, flap volumes, and flap surfaces.
Innervation is restored in skin grafts by the regeneration of nerve endings from the edges of the defect and from the graft bed [
15,
20]. Therefore, an innervated bed is required to regain innervation [
20]. Furthermore, skin grafts are mostly innervated by unmyelinated fibers, which transmit perceptions of temperature, pain, and gross touch, whereas finer sensations (e.g., light touch, vibration, and the 2-PD examination) are transmitted by heavily myelinated fibers and specialized sensory receptors [
15].
This might partially explain why finer sensations can be partially restored. However, protective qualities remained poor in our study.
The regeneration of autonomic fibers along the arterial pedicle and into a free flap is another potential mechanism of sensory reinnervation in non-neurotized free muscle flaps, which may explain the better SW outcomes in zone 1 [
21]. Furthermore, the thickness of the skin graft might be of particular relevance. Full-thickness skin grafts contain target structures (e.g., hair follicles, erector pili muscles, and sweat glands), and can potentially regain reasonable autonomic innervation. Skin receptors in full-thickness skin grafts exert a chemical attraction for the axons of the regenerated nerve, a phenomenon known as neurotropism [
22]. However, in split-thickness skin grafts, these structures are lacking, and full-thickness skin grafts might be superior to partial-thickness skin grafts [
9].
Our study has limitations, such as a limited sample size with a relatively short follow-up period, a lack of homogeneity, and the omission of a formal control group for comparison of our results. In addition, while there are myriad choices of free muscle flaps, such as the latissimus dorsi muscle flap, the serratus anterior muscle flap, and the rectus abdominis muscle flap [
8], we only evaluated the free gracilis flap with a meshed skin graft after lower extremity reconstruction. Therefore, prospective randomized controlled trials are needed, including measurements of painful protective sensations by two independent investigators and correction for covariates, to confirm our findings. Moreover, anatomical and histological studies are needed to assess the origin of the partial spontaneous return of sensitivity to skin grafts over muscle flaps.