

Ethical approval
The study was approved by the Institutional Review Board of Stanford University Medical Center (IRB No. IRB-44367) and performed in accordance with the principles of the Declaration of Helsinki. Written informed consents were obtained.
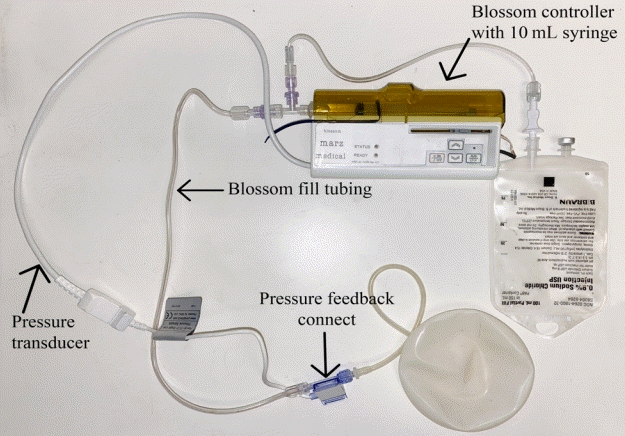
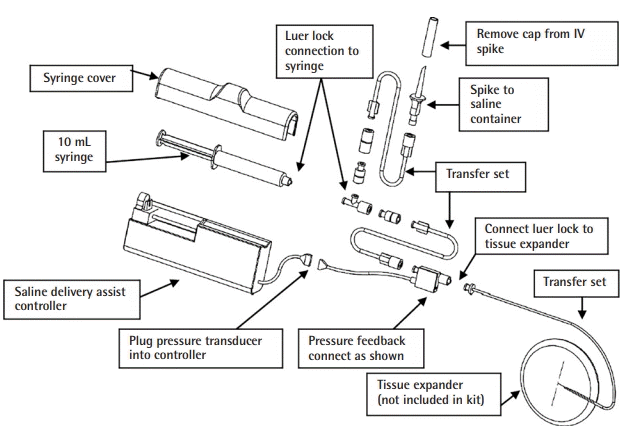
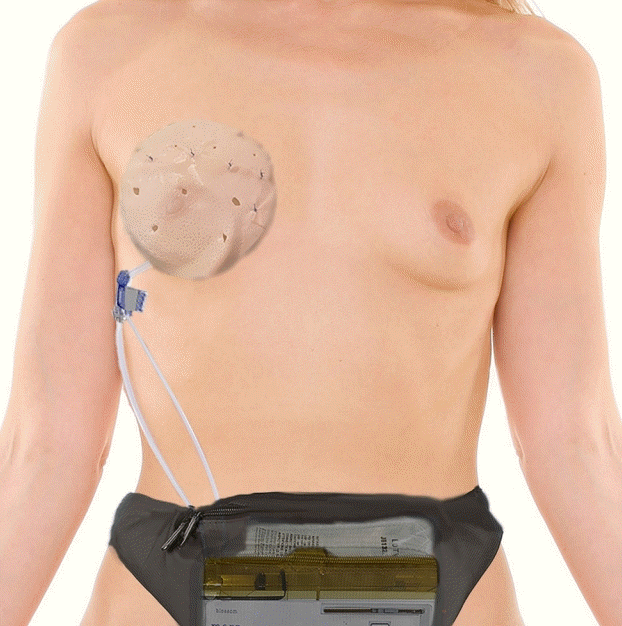
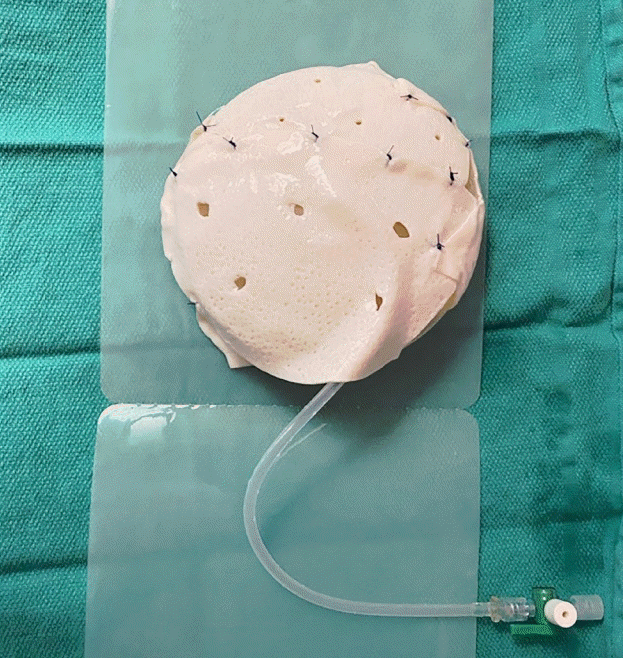
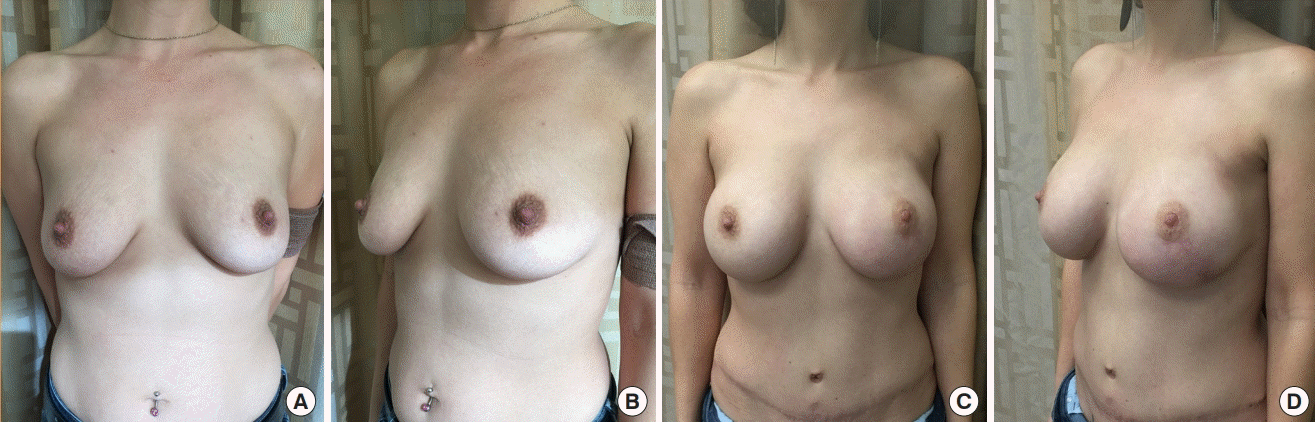

| Variable | Value |
|---|---|
| No. of tissue expanders | 22 |
| Age (yr) | |
| Mean ± SEM | 43.36 ± 0.10 |
| Median | 42 |
| 25th – 75th percentile | 35.0–48.8 |
| BMI (kg/m2) | |
| Mean ± SEM | 23.8 ± 1.1 |
| Median | 21.95 |
| 25th – 75th percentile | 19.93–27.78 |
| Variable | No. of breasts (%) |
|---|---|
| Prior radiation therapy | 6 (27.3) |
| Neoadjuvant chemotherapy | 10 (45.5) |
| Adjuvant chemotherapy | 2 (9.1) |
| Never-smokers | 19 (86.4) |
| > 12 pack-year smoking history | 3 (13.6) |
| Statistical analysis | Flap with implant (day) | Implant only (day) | P-value |
|---|---|---|---|
| Mean ± SEM | 13.4 ± 1.3 | 11.7 ± 1.4 | 0.78 |
| Median | 12 | 13 | |
| Range | 8–23 | 8–16 | |
| Interquartile range | 9.0–16.5 | 8.0–14.5 |
The flap with implant column includes patients who underwent flap and implant-based breast reconstruction. The implant only column includes patients who underwent implant-based breast reconstruction. Time to full expansion was defined as the number of days from initiation of saline infusion until complete delivery of the total programmed volume.
SEM, standard error of the mean.
The P-value was calculated using the paired t-test, and statistical significance was set at P<0.05.
Major complications included infections resulting in tissue expander (TE) removal and any issues requiring surgical intervention and/or hospitalization (e.g., infection requiring intravenous antibiotic therapy, wound closure revision, surgical drainage of expander pocket, and TE explantation). Minor complications included issues that did not require surgical intervention (e.g., hematoma, seroma, infection requiring oral antibiotics, delayed wound healing, and uncontrollable pain). The flap column includes patients who underwent flap and implant-based breast reconstruction. The implant column includes patients who underwent implant-based breast reconstruction.
The flap column includes patients who underwent flap and implant-based breast reconstruction (n=15). The implant column includes patients who underwent implant-based breast reconstruction (n=7).
BMI, body mass index; NSM, nipple-sparing mastectomy; NNSM, non-nipple-sparing mastectomy; Y, yes; N, no.
Percent (%) and P-values reflect the two separate groups. The Fisher exact test was performed to determine two-tailed P-values. Statistical significance was set at P<0.05.