INTRODUCTION
Mucormycosis is a rare, life-threatening fungal infection that typically progresses rapidly [
1-
3]. It is caused by filamentous fungi belonging to the
Mucorales order; the pathogens most commonly responsible for human disease are
Rhizopus species (spp.) [
1-
4]. Other human pathogens include
Mucor,
Cunninghamella,
Absidia,
Lichtheimia, and
Apophysomyces spp. with greater than 20 human pathogens identified [
1,
4]. Prior literature estimates the annual incidence of mucormycosis is less than two cases per million individuals [
3]. There is evidence to support an increasing incidence of mucormycosis as the population at-risk has grown significantly [
3,
4]. However, it remains difficult to determine the true incidence of mucormycosis as many suspected cases lack definitive mycologic or histologic evidence [
1,
3,
4].
Patients at-risk of mucormycosis are typically immunocompromised especially those with uncontrolled diabetes, hematologic malignancies, and undergoing solid organ or hematopoietic stem cell transplantation [
2,
3]. Immunocompetent patients experiencing major penetrating trauma and burns are also predisposed to mucormycosis [
2,
3]. Most infections are community-acquired and occur via inhalation of fungal spores or direct inoculation via violations in the cutaneous barrier [
2,
3]. Reports of hospital-associated mucormycosis are becoming more common; contaminated catheters, dressings, and oral medications have been implicated [
4]. Mucormycosis is divided into six forms based on the anatomic site involved: rhinocerebral, pulmonary, cutaneous, gastrointestinal, disseminated, and uncommon presentations [
2]. All forms are characterized by angioinvasion, which manifests clinically as vessel thrombosis and tissue necrosis, and carry a high risk of mortality [
2]. In the diabetic population, most commonly affected by rhinocerebral mucormycosis, the mortality rate is reported to be 44% [
3].
Cutaneous mucormycosis, the third most common form, often affects immunocompetent patients [
4]. Only 10% to 15% of cases are reported to involve diabetic hosts [
4]. The most common risk factor, implicated in up to 88% of cases, is penetrating trauma with soil being the source of inoculation [
2,
4]. Burns, surgery, persistent maceration, and subcutaneous or intramuscular injections are also risk factors [
2,
4]. Cutaneous infection can remain localized, invade deeper structures (e.g., muscle, tendons, and bones), or disseminate to other organs [
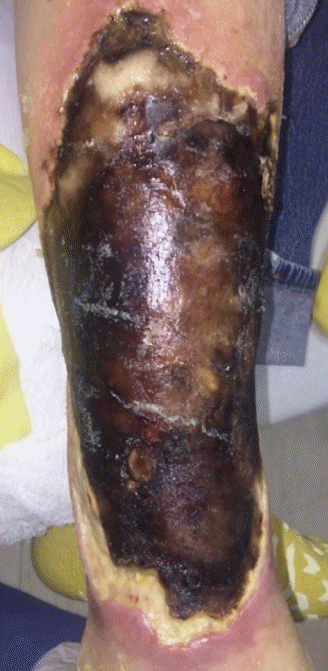
4]. When deep extension occurs, it usually presents as necrotic eschar with surrounding erythema and necrotizing infection [
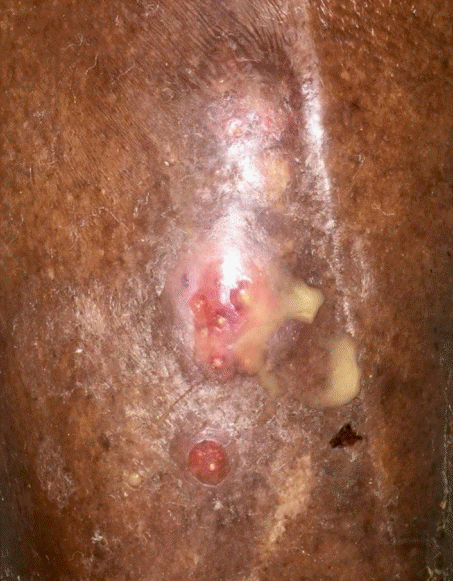
4]. However, cutaneous mucormycosis may also present indolently with skin findings limited to a small erythematous macule [
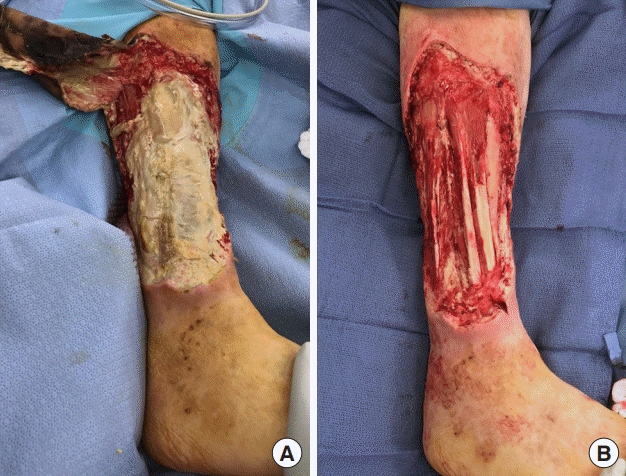
3]. Urgent, aggressive surgical debridement is the cornerstone of treatment for all presentations of cutaneous mucormycosis [
2]. Prompt diagnosis, control of predisposing factors, and appropriate antifungal treatment are also essential [
2]. We report two cases of lower extremity cutaneous mucormycosis with deep extension in diabetic patients. These are the first reported cases of cutaneous mucormycosis resulting in amputation in the absence of major penetrating trauma, recent surgery, or burns. Institutional Review Board approval was obtained (IRB No. 2018-173) for review of patient records. Both patients provided informed consent for the publication of their clinical case and the accompanying preoperative, intraoperative, and postoperative photographs.
DISCUSSION
Cutaneous mucormycosis remains uncommon with less than 200 reported cases identified in a recent meta-analysis [
7]. Of those, deep extension occurred in only 37 cases. Limb loss in the setting of cutaneous mucormycosis is even less common. To date, there have been three reports of progression to amputation; all of these cases involved significant disruptions in the cutaneous barrier in the form of trauma, surgery, or burn wounds [
8-
10]. However, our cases are unique because deep extension of mucormycosis and limb loss was preceded by seemingly mundane superficial wounds. There was no history of recent surgery or trauma which would have facilitated inoculation of deeper structures. Challenges in diagnosis and the highly comorbid nature of these patients both contributed to the poor outcome. Despite this, both patients healed well without further complications and received prosthetics at 4 months after their amputation.
In our first case, mucormycosis presented with classic findings of necrotic eschar and erythema. Light growth of
Rhizopus was identified on day 6 of admission. However, the likelihood of
Rhizopus as a contaminant and the risk of nephrotoxicity delayed initiation of amphotericin B until day 10 of admission. Progressive necrosis and inability to achieve clean margins necessitated amputation. This is similar to the case reported by Stanistreet and Bell [
8] in 2017, where widespread necrosis of the lower extremity developed after infection of a burn wound. In that case the diagnosis of mucormycosis was delayed for over 40 days, by which time the patient had globally deteriorated and subsequently expired despite the initiation of antifungal therapy.
Our second case had an atypical presentation with skin findings limited to a small ulcer with new purulent drainage. However, deep extension to the tibia had already occurred. Such a discrepancy in severity between cutaneous findings and overall extent of mucormycosis has been reported to occur in immunosuppressed patients [
3]. In this case, diagnosis of mucormycosis was made on day 11 of admission spurring a change in antifungal regimen from voriconazole to posaconazole as
Mucorales is reported to be resistant to voriconazole [
11]. Unexpectedly, final sensitivities revealed complete resistance to posaconazole but reasonable sensitivity to voriconazole. In a prior case of tibial osteomyelitis resulting from contiguous spread of mucormycosis from a pretibial ulcer, limb salvage was successful after surgical debridement and treatment with amphotericin B [
12]. The delay in effective antifungal coverage likely contributed to residual mucormycosis which necessitated more proximal amputation.
Delays in diagnosis and adequate treatment were significant factors in both cases. The relatively insidious nature of both presentations also resulted in a delay in seeking care. In fact, one of the infected ulcers was noted at a routine follow up appointment for a contralateral below knee amputation. Diagnosis traditionally relies on both cultures and histopathology [
6,
11]. When a patient comes in with a chronic purulent wound, fungal cultures should be sent immediately. Fungus on wound cultures may be an environmental contaminant; thus, correlation with histopathology is essential to confirm fungus as a pathogen [
6,
11]. Fungal cultures can also be falsely reassuring as they are only positive in 50% cases confirmed by histopathology [
11]. Given the need to confirm positive fungal cultures with histopathology, it is imperative that tissue samples for culture and histopathology be collected from the same area [
6]. This avoids inadvertently sampling a second area without fungal elements, which if sent to histopathology risks incorrectly concluding a positive fungal culture is due to contamination.
Early diagnosis and treatment are vital to decrease morbidity and mortality associated with mucormycosis; research has focused on new diagnostic methods [
11]. Molecular based techniques, such as polymerase chain reaction, can be utilized for detection and identification of
Mucorales [
1,
11]. Most mucormycosis pathogens are identified based on morphology; however, molecular studies are necessary for accurate identification of some species [
1]. Morphologically indistinguishable
Lichtheimia isolates previously thought to all be L.
corymbifera were determined to be two unique species based on divergent internal transcribed spacer sequences [
1]. Use of internal transcribed spacer sequencing to confirm morphologic identification of L.
corymbifera in the clinical setting has been described in a recent case report [
13].
The clinical utility of such molecular assays may be limited as prior literature reported no significant difference in antifungal sensitivities of
Lichtheimia spp. requiring molecular studies for speciation [
1]. However, the unusual antifungal sensitivities of L.
corymbifera in our second case demonstrate the value of molecular studies as faster speciation would have significantly altered clinical management. A recently published study on species-specific antifungal susceptibility profiles found the MIC of posaconazole for eight L.
corymbifera isolates was 0.1; thus, we believe this pattern of posaconazole resistance has not been previously reported [
14]. In the first patient’s case, sensitivity assays were not performed as LAmB, which is the drug of choice for mucormycosis, was utilized. However, susceptibility to amphotericin B does vary between species [
14]. In a 2014 case report, cutaneous mucormycosis caused by an isolate of
Apophysomyces with a high MIC to amphotericin B led to the patient’s death due to nonresponse to amphotericin B and fluconazole [
15]. Sensitivities may also have been of value in selecting a second antifungal agent when disease progression continued despite appropriate management.
Given the unprecedented aspects of these cases, mucormycosis warrants increased clinical suspicion, especially in diabetic wound patients with atypical presentations. In such patients, it is obligatory to send specimens for acid-fast bacilli and fungal cultures. It remains unclear whether the poor outcomes we report represent a “perfect storm” of host factors and diagnostic delays or if healthcare-associated Mucorales with increased virulence may have contributed as both patients had recent, prolonged exposure to healthcare facilities. Despite remaining an uncommon disease entity, increased awareness of the spectrum of potential presentations as well as the evolving nature of mucormycosis diagnosis and management is critical to minimize morbidity and mortality.