INTRODUCTION
External ear reconstruction for congenital microtia remains challenging. Staged reconstruction using autologous costal cartilage has been considered the gold standard for achieving reliable outcomes and low complication rates. The two-stage Nagata technique [
1-
4] is one of the most common methods. It involves implantation of a rib cartilage framework with lobule repositioning in the first stage. Elevation of the reconstructed auricle occurs in the second stage.
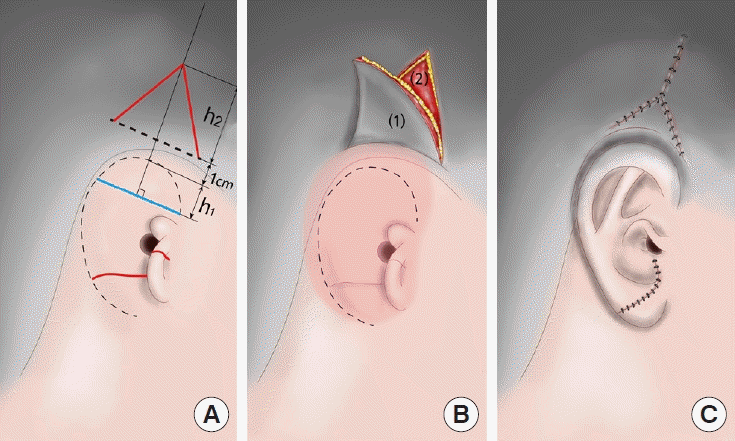
During the first stage of the operation, a thin pliable skin envelope and an elaborately sculptured three-dimensional cartilage framework are required to achieve a satisfactory convoluted shape. When preparing the subcutaneous pocket for the cartilage framework, a sufficient skin envelope can easily be obtained using adjacent tissue. This includes remnant vestiges. However, in patients who have previously undergone surgical procedures such as canaloplasty at a location suitable for the cartilage framework graft, it is difficult to acquire a pliable skin envelope due to postoperative scar contracture or tissue deficiency.
Inserting the fabricated cartilage framework without resolving this issue can result in tightening of the overlying skin envelope. This can lead to unsatisfactory convolution or even necrosis. To avoid these problems and obtain additional soft tissue, a tissue expander is frequently inserted prior to the first stage of cartilage framework implantation [
5]. However, this technique adds another surgical stage that requires general anesthesia. For unilateral microtia, this method requires at least three operations. Furthermore, to complete reconstruction for bilateral microtia, six operations are required. These multiple operations could represent a considerable burden for pediatric patients and their families, and they also pose a risk of foreign body-related complications, including infection.
A V-Y advancement flap is a traditional, simple procedure used to obtain additional soft tissue from surrounding areas. It has been used extensively for various types of reconstruction. For ear deformities, this method has played a key role in the correction of cryptotia [
6-
8]. However, it has not been used for the reconstruction of congenital microtia. We hypothesized that in patients with microtia and insufficient regional soft tissue, the V-Y advancement technique could yield an additional skin envelope. This technique is simple, reliable, and could provide satisfactory convolution.
DISCUSSION
In patients with congenital microtia who have undergone previous canaloplasty, the traditional strategy for reconstruction has been to use tissue expansion to overcome the issue of regional soft tissue deficiency. However, this has the limitation of adding reconstruction stages and associated patient morbidity. There have been certain trials to replace this method with postauricular skin flaps. Kim [
11] introduced a novel skin flap method using an extended scalp postauricular and isolated conchal flap. Hwang et al. [
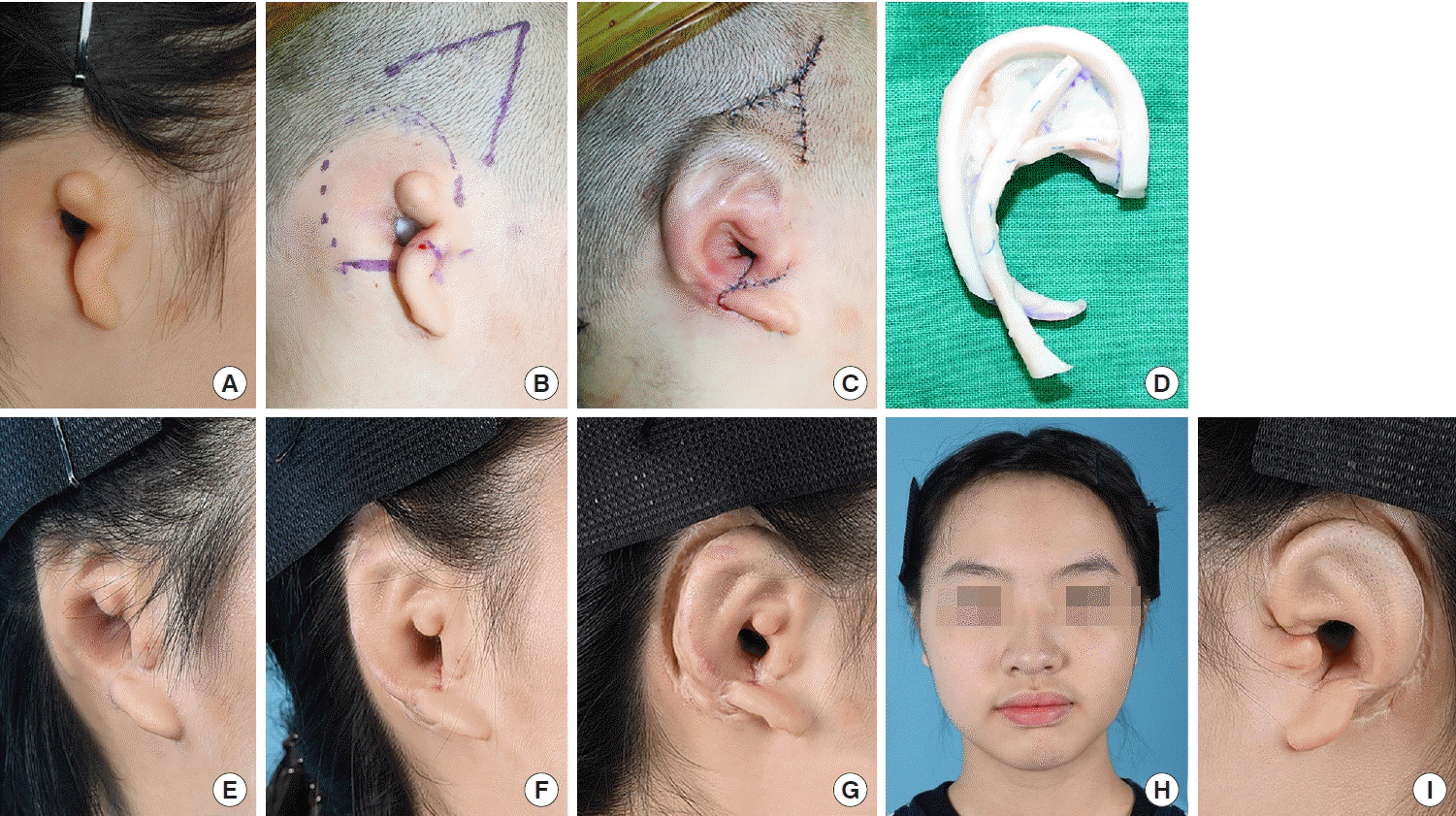
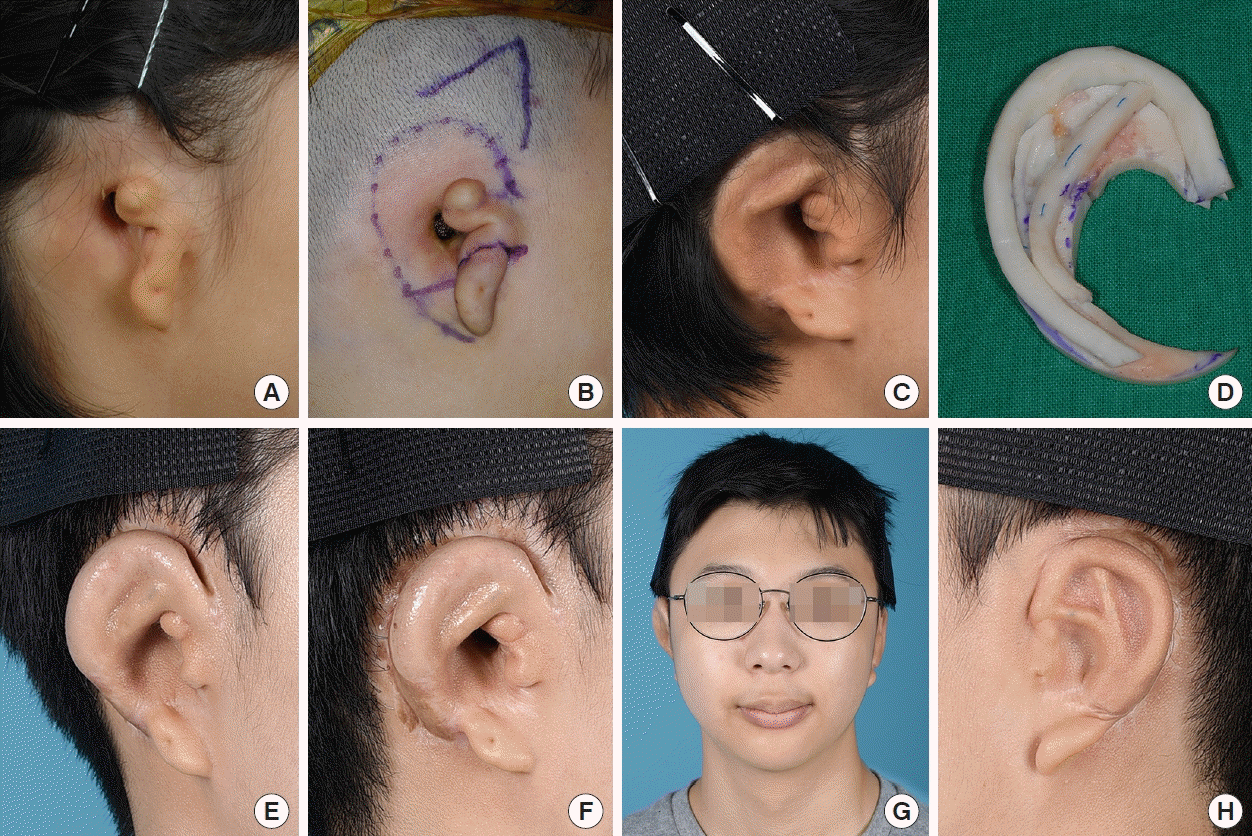
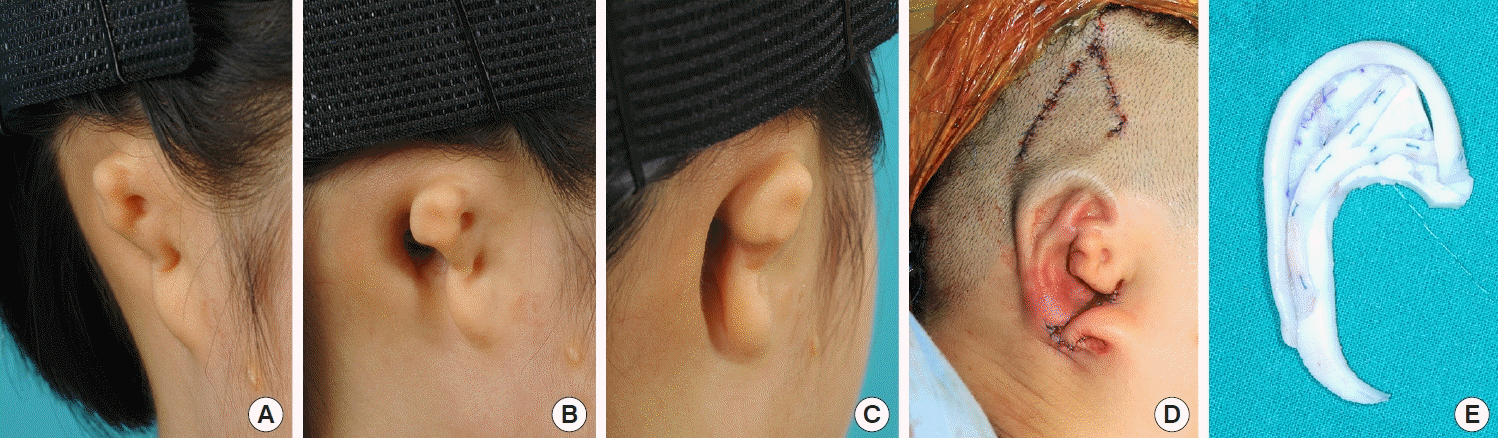
12] modified this method by extending the conchal flap and applied it to patients who had undergone previous canaloplasty. The results were satisfactory. However, this procedure required additional effort. Numerous elevated flaps were required as well as resurfacing with skin grafts from donor sites. In addition, there may be a potential risk of flap congestion, as multiple flaps converge on the embedded framework. In the current study, we adopted a simple method of V-Y advancement of a triangular temporal flap for reconstruction in eight microtia patients with challenging presentations. This method enabled us to obtain sufficient soft tissue, effectively reducing the number of operation stages and facilitating esthetically pleasing outcomes with a low complication rate.
As ear canal operations lead to vascular injury and scarring in the conchal region [
7], many attempts have been made to minimize the undesired impacts of auricular reconstruction. This has been accomplished by adjusting the timing of each reconstruction stage. Roberson et al. [
13] reported a higher meatal stenosis rate when atresia repair was conducted prior to Medpor insertion than when atresia repair was performed after microtia reconstruction. Chen et al. [
14] introduced a new flap technique for combining canaloplasty simultaneously with total auricular reconstruction by performing Medpor framework insertion. This showed favorable cosmetic results with acceptable complication rates. At our institution, patients with unilateral microtia were routinely sent to otologists at the end of the first stage of hearing evaluation. We recommend canaloplasty prior to auricular elevation if the expected success rate of hearing restoration is over 90%. Patients usually have functional hearing on the contralateral side [
10]. However, for bilateral microtia patients, hearing restoration is a major concern that must be addressed to prevent delays in speech and language development [
15]. Considering that we routinely use autogenous ribs for auricular construction, surgery should be postponed until rib growth provides sufficient cartilage for fabrication. Therefore, most of the patients underwent ear canaloplasty prior to auricular reconstruction. Thus, in this study, we focused on patients who had bilateral microtia with a history of canaloplasty prior to auricular reconstruction. We reviewed our experiences with microtia reconstruction using V-Y advancement of a temporal triangular flap in these cases and evaluated the outcomes.
Kubo [
8] first reported a surgical method using a V-Y advancement flap for cryptotia correction. The V-Y advancement flap provided sufficient skin to the upper and posterior portions of the auricle, and was also easy to apply in almost all cases by regulating the height of the triangular flap [
6]. In this study, we adopted a V-Y advancement technique for microtia reconstruction in cases with unfavorable regional conditions due to prior canaloplasty. Using this method, we achieved satisfactory results with respect to the costal cartilage graft and the auricular elevation. We also found that this method could be used safely without serious complications that could require revisional cartilage grafting. An imbalance between the skin envelope and the framework results in poor auricular definition. Moreover, a poorly vascularized skin envelope can result in skin necrosis, framework exposure, or long-term delayed cartilage resorption [
16]. Considering these aspects, the use of the V-Y advancement flap could provide enhanced stability compared to the tissue expansion method. The V-Y advancement flap could keep its own vascularity with adequate flap thickness. In addition, our method can achieve direct visualization with only a V incision on the premise that sufficient dissection with meticulous hemostasis of the future implantation site and proper traction will be conducted. After the implantation of the cartilage framework, negative-pressure suction was applied while the skin flap was temporarily fixed. We could then ascertain whether the cartilage framework was in the proper position and whether the flap supplied ample skin. We could adjust the framework until we achieved desirable results, thereby guaranteeing the security of the embedded cartilage framework without an additional incision.
Compared to the tissue expansion technique, the V-Y advancement technique does not pose a risk for capsular contracture. Capsular contracture can lead to ambiguous definitions and can contribute to stenosis of the newly created external auditory canal (EAC). In addition, this technique is free of risk for foreign body-related infections, which can result in EAC stenosis. Furthermore, various strategies to prevent further EAC narrowing after auricular reconstruction have been adopted. We attempted to maintain the dermal and subdermal plexus. We also included a subcutaneous pedicle in the skin flap to ensure sufficient blood supply. A history of hematoma could result in EAC stenosis. Therefore, surgeons must be vigilant regarding bleeding. Careful hemostasis and proper drain placement must be conducted. If canaloplasty is planned after a rib cartilage graft, a wide conchal bowl is created at the framework fabrication stage. This provides space for future tissue contraction and canal stenosis [
10]. A sufficient interval between operations (with an ideal time of at least 1 year) was also allowed to facilitate recovery. In addition, after ear canaloplasty, a customized ear mold was worn by the patient to prevent canal narrowing.
There are concerns regarding hair distribution when using the V-Y advancement flap method. When the triangular flap is inferiorly advanced, the hair-bearing portion of the flap base moves to the helical sulcus or possibly to the upper portion of the helix with a lower hairline. This can be successfully managed using laser epilation. We used a long-pulsed alexandrite laser system (GentleMax Pro, Candela Co., Wayland, MA, USA) under topical anesthesia with 5% lidocaine cream. The irradiated region was within 2 cm of the outline of the cartilage framework implantation site, as determined by the amount of lowered hairline. The irradiation fluence was 18–22 J/cm2 (3 ms, spot size 12 mm). In all cases, laser epilation was conducted after the rib cartilage graft. However, the optimal timing of laser epilation, either before or after auricular elevation, depends on the patient’s needs. We performed irradiation at least 2 months after the previous operation. The irradiation interval was every 4–6 months. If patients were satisfied with their appearance without additional irradiation, we completed the epilation procedures and encouraged patients to visit the clinic as needed. There were no side effects such as inflammation, scarring, skin contracture, or circulatory failure related to irradiation. With respect to donor site morbidity, a Y-shaped scar was inevitable. However, the scar can be hidden by hair growth.
Although our method using temporal V-Y advancement could provide sufficient soft tissue in the first surgical stage, and the rib cartilage graft demonstrated excellent convolution, concerns persist regarding the perfusion of the cartilage framework implantation site or the advanced triangular flap. In the first stage of the operation, we designed the lower border of the temporal triangular flap 1 cm from the designed helix. By distinguishing the flap from the cartilage framework, more perforators would be included in the flap. It also protects flap from the threat of ischemia. In the presented cases, we observed temporary ischemic signs on the framework implantation site immediately after rib cartilage implantation. However, these signs resolved quickly with conservative management. In the second stage of the operation, the transposed flap, the margin of which was surrounded by scar tissue, was elevated to harvest the superficial temporal fascia. The flap potentially could have had insufficient tissue perfusion, which could have led to necrosis. However, in the present study, the majority of patients had an uneventful course after the second surgical stage. Only one patient had partial necrosis of the transposed triangular flap. It is likely that, with its elaborate design that considered the incision line of the second surgical stage as well as the meticulous technique involved, our method would not result in adverse outcomes following the second surgical stage.
The method described in this study exhibited satisfactory esthetic outcomes in all patients and produced stable reconstructive outcomes. However, as we included only a small number of cases, the final outcomes of this technique require confirmation through further clinical research.