DISCUSSION
Kerrigan and Daniel [
4] studied the anatomy and course of the intercostal artery, which links the internal mammary artery with the aorta and forms an arcade. Throughout its course, the intercostal artery forms the vertebral, intercostal, intermuscular, and rectus segments. Among these four segments, the intercostal segment is the longest (approximately 12 cm) and gives rise to 5-7 musculocutaneous perforators. The diameter of the dominant intercostal artery perforator is approximately 0.8 mm, which is suitable for microscopic anastomosis. After Kerrigan and Daniel's report, few anatomical studies were published on intercostal artery perforators until Badran et al. [
5] conducted a study on dominant perforators arising from the mid-axillary line. Recently, Hamdi et al. [
1,
2] reported a more detailed study on lateral intercostal artery perforators. They classified these perforators as dorsal, lateral, and anterior artery perforators [
2]. Further, they statistically investigated the location of perforators in a cadaveric study [
1].
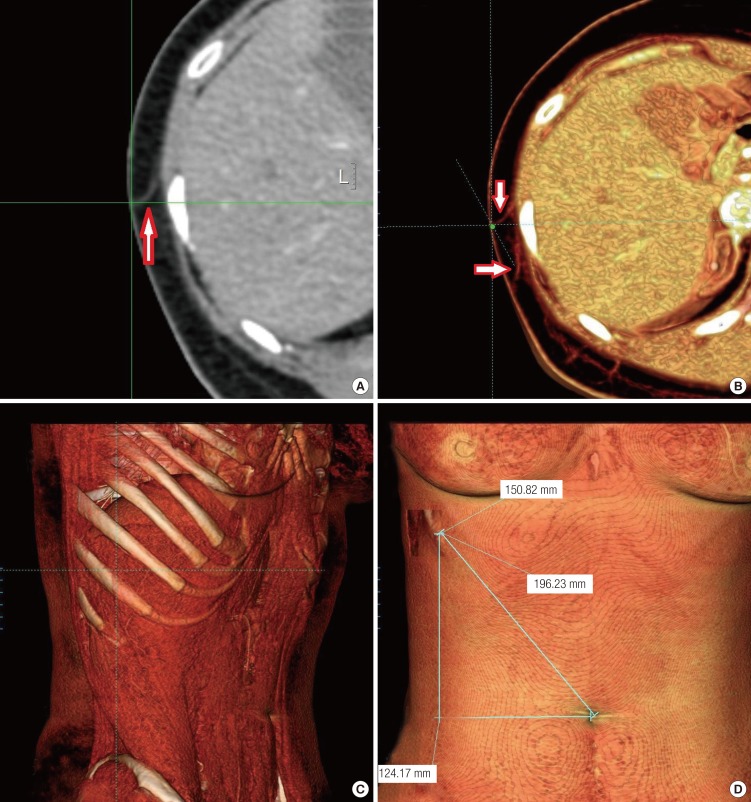
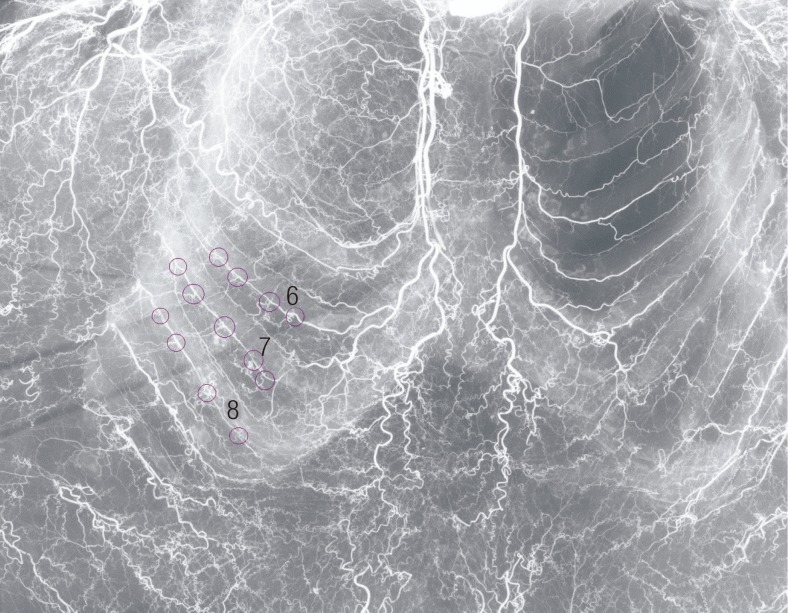
We performed angiography on a fresh cadaver and dissected the lateral intercostal artery perforators in another cadaver. Angiography in a fresh cadaver revealed many intercostal artery perforators, with more than four perforators and the dominant perforators located in the sixth to eighth intercostal spaces (
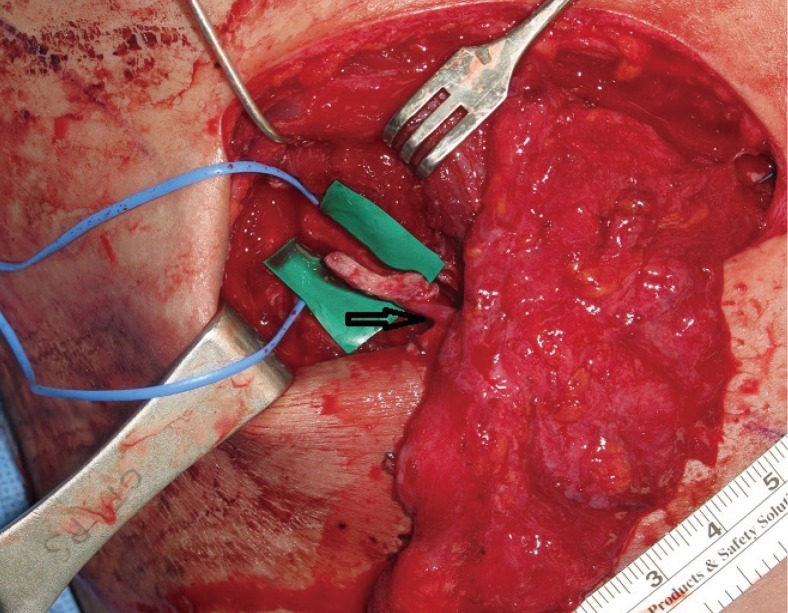
Fig. 7). Two lateral intercostal artery perforator-based adipofascial free flaps were elevated from the cadaver. Through the flap dissection, the pedicle length, diameter, and location of the perforator were evaluated. The pedicle diameters of the two flaps were 1.3 mm and 1.5 mm at the origin of the intercostal artery perforator. At the suprafascial plane, the perforator emerged from the serratus anterior muscle, 3 cm in front of the anterior border of the latissimus dorsi muscle. The pedicle lengths were 52 mm and 55 mm from the intercostal muscle fascia, and a further pedicle length of up to 65 mm was achieved with intercostal muscle dissection.
According to an anatomical study by Hamdi et al. [
1], the most reliable perforator of the lateral intercostal artery originated from the sixth intercostal artery. However, we used a flap from the seventh or eighth intercostal artery perforator, as it had sufficient pedicle diameter and length for anastomosis with the superficial temporal artery. Moreover, it may be suitable for anastomosis with the facial artery [
13-
15]. Badran et al. [
5] reported that a single perforator was sufficient to support a flap, even though many donor pedicles from the intercostal artery were available. They were able to elevate flaps as large as 25 cm×17 cm. In our study, a single pedicle provided sufficient nourishment for the survival of an entire flap. However, multiple pedicles were encountered during flap dissection. If we had inadvertently cut one pedicle, we could have used another.
The largest flap that we used was 15 cm×7 cm, but it developed partial fat necrosis. Therefore, it appears that an adipofascial flap can be safely elevated at a maximum size of approximately 12 cm×7 cm; however, additional clinical studies are needed to confirm this observation. We believe that the vertical length of the adipofascial flaps should not exceed 10 cm. If the vertical length of these flaps is greater than this value, the circulation of the skin flap at the donor site may be affected. Although we experienced partial failure with a 15 cm×7 cm flap, the donor site healed without problems such as seroma or hematoma [
16]. To avoid donor site morbidities, we elevated the adipofascial flap below Scarpa's fascia. No skin necrosis or dehiscence at the flap donor site was observed with this technique, and minimal scarring was observed.
A common procedure for correcting facial depression is fat injection [
16-
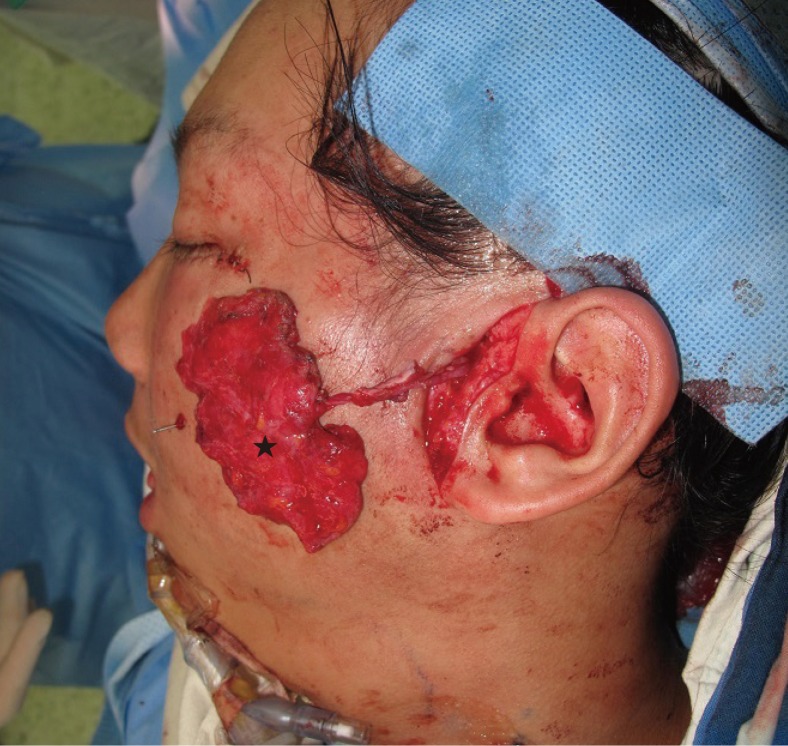
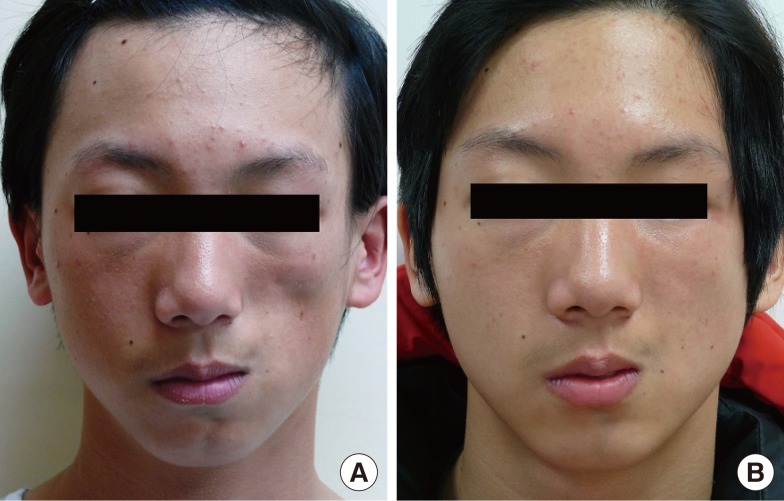
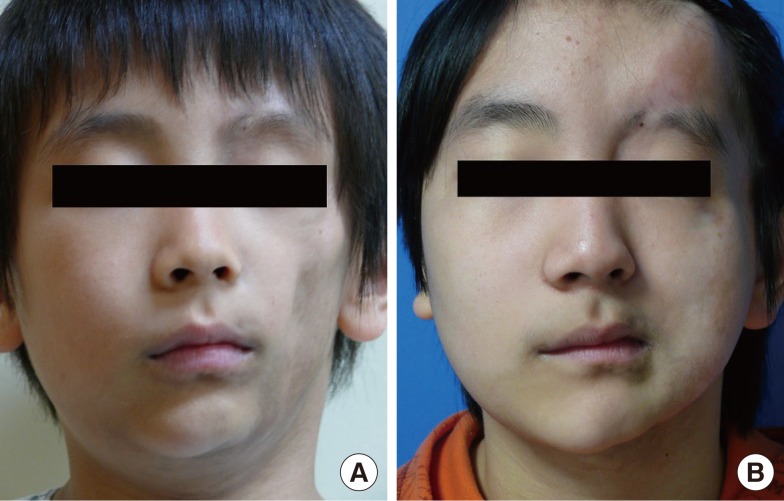
18]. Many plastic surgeons inject free fat at the depression sites, and this procedure is usually repeated multiple times. However, one of our patients with hemifacial atrophy had considerable tissue deficiency, and we could not achieve facial symmetry using fat injections alone. In this case, the lateral intercostal artery perforator-based adipofascial flap was able to cover the volume deficiency. When we needed additional volume support after disease progression, we used dermofat from the previous donor site.
The previous literature concerning intercostal artery flaps mainly described procedures for breast and lower trunk reconstruction [
1,
6-
12]. To the best of our knowledge, despite the multiple benefits of lateral intercostal artery perforator-based adipofascial free flaps in facial reconstruction, no clinical studies on the efficacy of this type of flap have been reported. Various free flaps have been used for facial contour reconstruction, such as groin, omentum, scapular, and parascapular flaps; transverse rectus abdominis muscle flap; radial forearm adipofascial flap; and anterolateral thigh flap [
19-
24]. However, the lateral intercostal artery perforator-based adipofascial free flap may be the best option in terms of tissue quality, versatility, convenience, and decreased morbidity compared with other flaps.
First, the cheek is a commonly involved area of depression, and the texture of the cheek is usually soft. Therefore, a flap with soft texture that does not contain dermis is more suitable for cheek depressions. The lateral intercostal artery perforator-based free flap has thicker subcutaneous tissue; therefore, substantially more adipose tissue can be harvested compared with that in the case of the groin-free flap or superficial circumflex iliac artery-free flap. The deep inferior epigastric perforator flap has excess adipose tissue and has shown satisfactory results for the correction of facial atrophy [
25]. However, defatting evenly in the deep layer is problematic because the pedicle lies in this layer.
Second, a lateral intercostal artery perforator-based free flap has versatility because of its structural composition, which is composed of skin, subcutaneous dermal fat, fascia, and bone. The rib bone may be taken simultaneously, and it may be taken again through the existing scar. The reconstruction of bone as well as that of soft tissue is required in facial asymmetry, and the rib bone is most commonly used for bone graft. If the dermis is required for another area of reconstruction, it can be harvested separately from the flap elevation or can be harvested during a second surgery. Compared with a de-epithelialized flap, the donor site had less tension. Therefore, a dermofat graft can be obtained easily.
Third, harvesting the lateral intercostal artery perforator-based adipofascial free flap for facial reconstruction is convenient. If the pedicle for an anterolateral thigh free flap is a musculocutaneous branch, intramuscular dissection is time consuming because of the thickness of the vastus lateralis muscle. However, since the lateral chest wall has more thin muscle than the thigh, pedicle dissection could be performed with little effort. In addition, as mentioned before, the presence of more than one perforator in this flap afforded the ability to use another perforator if the first was accidentally damaged [
5]. Further, the two surgical fields are close to each other, and two surgical teams may approach without changing positions because the location of the lateral intercostal artery flap anterior to that of the thoracodorsal artery perforator flap, latissimus dorsi flap, or scapular or parascapular free flap. Moreover, because the lateral chest wall area has little skin tension, pedicle dissection can be performed through a linear incision with low effort retraction.
Fourth, the lateral intercostal artery perforator-based adipofascial free flap reduces donor-site morbidity. The donor site could be closed without skin tension because we did not remove the skin and the dermis. Therefore, the scars were more cosmetic. Moreover, the lateral chest donor site could be well concealed by wearing a sports bra or swimsuit, making it an even more favorable donor site for cosmetic and reconstructive surgeries. For example, the anterolateral thigh flap inevitably leaves visible depressed scars. The risk of pneumothorax, seroma, and hematoma should be noted. However, the incidence of pneumothorax was very low with careful management, if we did not harvest the rib bone or perform intercostal muscle dissection for pedicle lengthening. Since flap elevation does not disturb axillary lymphatics or involve dissection over a broad area like the latissimus dorsi free flap, the incidence of seroma and hematoma is also anticipated to be low. Furthermore, seroma and hematoma can be prevented by suction drainage and compressive dressing.
However, there are certain disadvantages to the use of lateral intercostal artery perforator-based adipofascial free flaps. In very thin patients, the adipofascial flap may be very difficult to elevate and a flap of sufficient thickness may be difficult to obtain. In such cases, a folded adipofascial flap can be used. Moreover, as the flap cannot be viewed, it is difficult to monitor the flaps. Therefore, the drainage patterns should be carefully assessed.
Facial asymmetry cannot be corrected with single-tissue grafts. Patients with facial asymmetry require a more complete procedure to restore their previous figure. The role of vascularized adipofascial free flaps used in our series is also very important. The utilization of rich vascular tissue from the adipofascial free flap provided the additional effect of restoring texture.
This is the first study on facial contour reconstruction using lateral intercostal artery perforator-based adipofascial free flaps. The use of this type of flap was effective and can be considered a good alternative for restoring facial symmetry in patients with severe facial soft tissue deficiency.