DISCUSSION
Infections caused by the RGM species are relatively uncommon. However, several studies have reported that outbreaks of a nosocomial surgical site RGM infection, most often due to
Mycobacterium abscessus or
M. fortuitum, have been associated with augmentation mammoplasty, liposuction, and facial plastic surgery [
2,
4,
5]. Clinical diagnosis of mycobacterial skin and soft-tissue infections is not easy to perform, and even when performed correctly, the diagnosis often takes a significant amount of time for the final confirmation. In fact, there have been instances in which delays of more than a year have been reported [
6]. Some of the key reasons for the difficulty of a speedy and accurate diagnosis are 1) high costs for the polymerase chain reaction test in order to detect NTM, 2) a relatively long time of 6-8 weeks for the mycobacterial culture incubation period, and 3) a lack of awareness of the possibility of NTM infections. Surgeons should recognize the possibility when caring for patients with chronic skin and soft-tissue infections following an invasive aesthetic procedure.
A series of case reports of a total of 88 patients of NTM soft-tissue infections had been widely reviewed in order to reveal common clinical history and disease courses among the patients. All patients in the literature had experiences of invasive procedures such as acupuncture, fat grafts, liposuction, and implant insertion. Interestingly, 70 patients (79.5%) suffering from uncontrolled infections were reported to visit a hospital within three months (range, 10 days to 12 weeks) after their first exposure [
3,
7,
8,
9,
10]. On the basis of the experiences reported in the literature, we formulated the clinical criteria consisting of one major and four minor factors that could discriminate the patients suspected of NTM infections from those with pyogenic infections. We assumed the history of invasive procedures within three months to be the key factor in our criteria. Even though a few cases reported the detection of a pathogen more than three months after the initial exposure, we determined three months to be a reasonable period to cover a majority of patients with NTM infections. The minor criteria reflected clinical symptoms, laboratory findings, and comorbidities that were commonly found among patients with NTM infections.
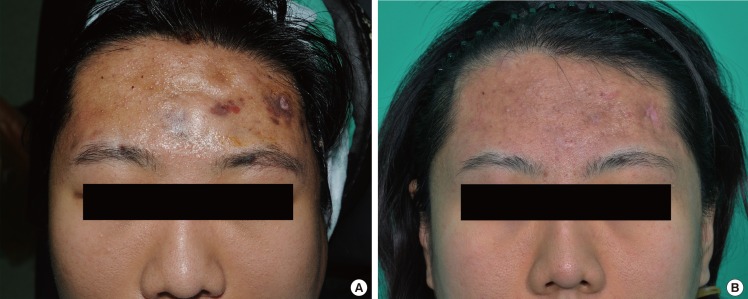
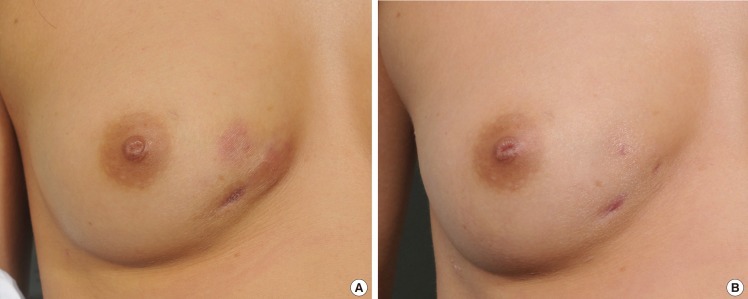
There have been numerous clinical and laboratory trials for the early confirmation of mycobacterial infections. Initially, we focused on patients infected surgically with atypical cutaneous manifestations localized at the sites of previous aesthetic procedures. According to our findings, skin and soft-tissue infections of NTM look slightly different from pyogenic bacterial infections. First, the color of erythema around the focal lesion is darker and more purplish than the typical reddish color of pyogenic induration. This is probably due to the fact that the natural course of an NTM infection is chronic, and the pathogen continually induces hyperpigmentation of the affected skin lesions [
11]. Second, NTM cutaneous infections seem more likely to form deep nodules with multiple deep sinus tracts containing fluids that seem to be more serous than the sticky yellowish pus observed in the case of bacterial infections [
12]. These were not critical features but played important roles in the identification of NTM infections.
The diagnosis of NTM can be also made on the basis of histopathological findings. The presence of central caseous necrosis with amorphous granular debris, loss of cellular detail, and the presence of Langhan's giant cells are sufficient characteristics to make a reasonably accurate diagnosis of a mycobacterial infection. Although a number of different diseases such as brucellosis, mycoses, actinomycosis, and syphilis are involved in the formation of granuloma, mycobacterial infection is the most common microbial cause of caseating granuloma [
13]. In the case of the histologic results that showed caseating granuloma, we considered these findings to be an NTM infection until the mycobacterial culture result was confirmed to be negative. The positive findings of the histopathological exam allowed us to make an earlier start of empirical treatments for NTM.
Universally, the AFB stain and culture have been widely used in the diagnosis of mycobacterial infections. Even though the AFB stain is known to be a rapid, convenient, and cost-effective method, it has low sensitivity, ranging from 22% to 78%, and is unable to discriminate NTM from
M. tuberculosis [
14]. Culture, on the other hand, demonstrates a higher sensitivity than an AFB stain, as well as nearly 100% specificity in the diagnosis of mycobacterial infections; however, this method requires an extended time of 6-8 weeks.
Recent advancement in mycobacteriology laboratory techniques plays a critical role in the laboratory diagnosis of tuberculosis and of diseases caused by NTM. Advanced PCR tests are now widely used in the early detection of NTM infections and species identification. These techniques include restriction fragment length polymorphism (RFLP), PCR-reverse blot hybridization assay (REBA), and real-time PCR. RFLP uses restriction enzymes to break DNA into fragments, which are then compared with one another to distinguish the difference in the DNA sequences. Although this technique is convenient, many variables can affect results because of the use of electrophoresis in the comparison of the DNA fragments [
15]. PCR-REBA utilizes an oligonucleotide probe that specifically binds to certain DNA sites of specific pathogens, enabling a rapid identification of the 19 most common mycobacterial species, including
M. abscessus,
M. fortuitum complex, and
Mycobacterium chelonae [
2]. Both the sensitivity of NTM detection and the specificity are 100% for the DNA from the cultured isolated specimens [
16]. The success rates for NTM identification obtained by targeting the 16S rRNA,
rpoB, and
hsp65 genes are 65%, 82%, and 87%, respectively [
17].
In this study, we used an AdvanSure TB/NTM real-time PCR kit (LG Life Sciences, Seoul, Korea) for the detection of mycobacteria. Real-time PCR both detects and quantifies the DNA as the reaction progresses. Despite its limitation in terms of the number of pathogens that can be identified in a single exam, this technique has high specificity and sensitivity, and allows a quantitative analysis of the pathogen. The AdvanSure kit collects the products from two simultaneous reactions: one reaction occurs between the probe and the
rpoB gene site of NTM or the
IS6110 site of the
M. tuberculosis complex, and the other reaction occurs when the TaqMan probe is broken down by hybridization. Due to the simultaneous use of primer and probe hybridization, AdvanSure has a sensitivity and a specificity of 100%. Other studies pertaining to sensitivity and specificity in relation to this product have reported a sensitivity of 92.7% and a specificity of 91.3% [
18]. In our institution, the cost of a real-time PCR test is eight times the cost of an AFB smear. However, as real-time PCR has the advantages of a higher detection rate and a shorter result waiting time of 2.3 days on average, we strongly recommended this test to all the patients. Despite our efforts, 3 out of 12 patients refused the PCR test because of its high cost.
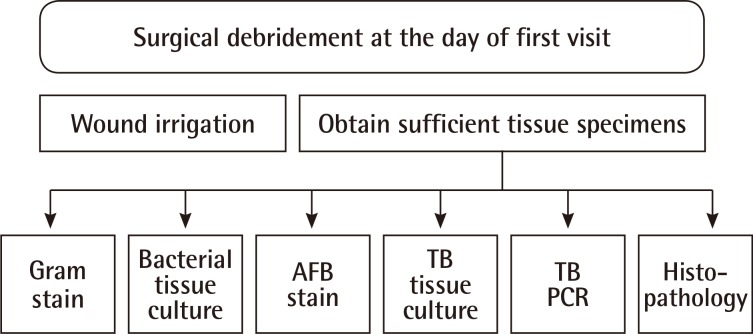
Throughout the study, surgical incision, drainage, and debridement were executed in the operating room at the patients' first visit. When performing a wide excision or drainage of abscesses under local anesthesia, more than 3 mL of the tissue specimen was collected in most patients in order to conduct various laboratory studies simultaneously. This was done under the assumption that a sufficient amount of tissue specimen will decrease the rate of false negative results. Moreover, infection wounds were washed with saline far more thoroughly and efficiently in the operating room.
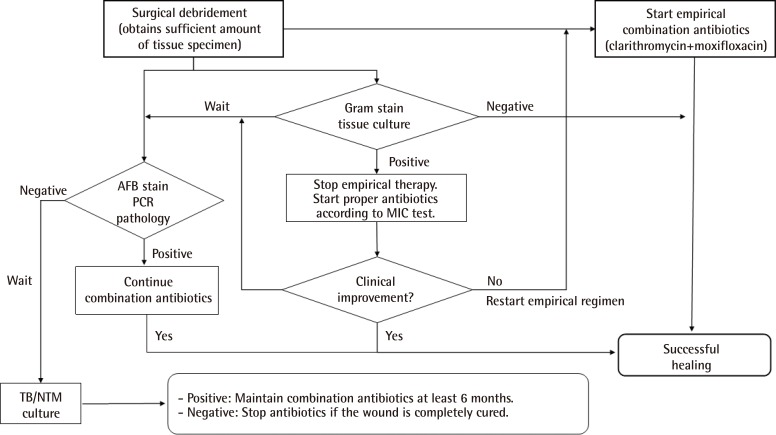
In earlier studies on the NTM treatment, empirical cephalosporin or ampicillin antibiotics were usually changed to anti-NTM medication including macrolides when obtaining laboratory evidence such as positive AFB staining or a positive PCR test [
2,
19]. Throughout this study, the focus was on the inclusion criteria of patients "highly suspected of NTM infection," which emphasized the previous history of aesthetic procedures, atypical cutaneous manifestations, and serial negative microbial tests. Thus, an empirical regimen of clarithromycin and moxifloxacin (Orally or IV) targeting the NTM infection was initiated from the day of the first visit without hesitation despite the absence of the NTM infection evidence at that time. As a result, the start date of empirical antibiotics could be pushed ahead by 2-3 days on the basis of these decisions.
Treatment guidelines published jointly by the American Thoracic Society and the Infectious Diseases Society of America (ATS/IDSA) are a valuable resource for the practicing clinician [
1]. These guidelines focus on initial management and emphasize that susceptibility testing should be performed on all mycobacterial isolates. It is known that an effective treatment of NTM skin infections includes surgical debridement along with a combination of antibiotic therapy. An antibiotic combination must be administered for a sufficiently long period of time to ensure complete wound healing and no recurrence. In order to prevent drug resistance and clinical relapse, antimicrobial therapy should be a multidrug regimen with a macrolide base as recommended [
1].
A combination of clarithromycin and moxifloxacin was selected for the empirical regimen decided on the basis of multiple consultations by the infectious disease expert at our institution. We administered the combination regimen to the patients while waiting for microbial evidence on whether there was a strong suspicion of an atypical mycobacterial infection. Clarithromycin emerged as the single best empiric choice for cutaneous NTM infections [
20]. Yu et al. [
19] reported that the rate of drug resistance to clarithromycin in the case of RGM was 12.5%. Rifampin, amikacin, imipenem, ciprofloxacin, and cefoxitin have all been used to good effect, provided that the disease was not at an advanced stage. Moxifloxacin is one of the most recently marketed fluoroquinolone antimicrobials. It differs from ciprofloxacin in having much better
in vitro activity against Gram-positive aerobes while retaining potent activity against Gram-negative aerobes. It is also active against the species of atypical mycobacteria associated with dermatologic infections. Moxifloxacin penetrates well into the inflammatory blister fluid and muscle and subcutaneous adipose tissues, which makes it a reasonable option for the treatment of skin and soft-tissue infections [
10]. Gillespie et al. [
21] evaluated the activity of moxifloxacin in comparison with that of four other quinolones and isoniazid against clinical isolates of mycobacteria. It proved to be the most active of the quinolones tested against
M. tuberculosis (MIC
90 0.25 mg/L),
Mycobacterium avium-intracellulare (MIC
90 1.0 mg/L),
Mycobacterium kansasii (MIC
90 0.06 mg/L), and
M. fortuitum (MIC
90 1 mg/L). Interactions between moxifloxacin and macrolides against clinical isolates of
M. abscessus and
Mycobacterium massiliense were also studied. The addition of moxifloxacin to macrolide therapy showed a treatment benefit to
M. massiliense in contrast to negative influences on the treatment outcome of the
M. abscessus infection [
22].
Since clarithromycin and moxifloxacin together have a wide range of antimicrobial activity, it is a reasonable choice among the wide range of empirical agents. We experienced a single case of an atypical
S. maltophilia infection followed by autologous fat grafting by repeated tissue cultures. The positive culture result was recognized 11 days after the start of the empirical treatment, and the pathogen was proven to be susceptible to moxifloxacin. The patient showed rapid clinical improvement within 2 weeks and continued oral moxifloxacin for 4 weeks despite the wound being completely healed. The efficient antimicrobial activity of moxifloxacin against
S. maltophilia was widely reported in the cases of meningitis and keratitis treatment [
23,
24]. Moreover, this regimen was effective in three patients who did not demonstrate any evidence of the pathogen. We assumed that the broad spectrum of clarithromycin and moxifloxacin activity covered certain pathogens that were not isolated or identified.
We continued to prescribe this regimen for 4 weeks to one patient having an
S. maltophilia infection and three patients showing no microbial evidence. This regimen was administered to three patients demonstrating caseous necrosis in histopathologic exams for at least 6 weeks until the mycobacterial culture was confirmed to be negative. Five patients confirmed to have an NTM infection were administered this regimen for 6 months. Although moxifloxacin and clindamycin were not always the best choice of antibiotics according to the susceptibility results, we decided to continue this regimen for the following reasons: First, all the five cases already showed complete wound healing state at the time when the susceptibility results were reported. Second, both macrolides and quinolones were demonstrated to be effective along with the minimum inhibitory concentration required to inhibit the growth of 90% of organisms (MIC
90), of less than 0.5 mg/L against the pathogens in all the cases. Moreover, the infectious disease expert expected the benefit of maintaining this regimen to far surpass the risk of adverse effects caused by antibiotic changes. Eight patients with caseating necrosis or NTM confirmation were followed up by the infectious disease expert as well. Most cases demonstrated clinical improvement within 2 weeks, and wound healing within 4 weeks after the first visit. Recurrence and any adverse effects associated with antibiotics were not observed during the follow-up period. The optimal length of therapy is unknown but is likely to be shorter than that in the case of a pulmonary or disseminated disease; current recommendations by ATS are for 6-12 months [
1].
The empirical treatment prior to pathogen identification was effective for the highly suspicious group of NTM patients in terms of reducing the time taken for clinical improvement. Moreover, a regimen of a combination of clarithromycin and moxifloxacin demonstrated excellent treatment outcomes of NTM, atypical bacteria, and local inflammation of unknown causes. Simultaneous execution of laboratory tests also saved time in identifying the pathogen. This empirical treatment strategy was based on multiple clues from the history and clinical manifestations and provided an efficient option for the management of skin and soft-tissue infections as compared to the standard management.