INTRODUCTION
When tissue is disrupted in vertebrates, the wound healing process begins, which is generally divided into three phases: inflammation, proliferation, and maturation [
1]. Each phase is regulated by cytokines, growth factors, and their cellular receptors [
2]. In this study, we focused on 1) collagen synthesis and 2) erythema caused by angiogenesis during wound healing.
First, collagen is a main protein of human skin, allowing for much of the skin's remodeling and turnover [
3]. Skin fibroblasts generate a precursor molecule called procollagen, which is converted into collagen [
4,
5]. Previously, collagen was thought to act only as a structural support. However, it has been reported that collagen is a major factor of wound healing. There are two important regulators of collagen production: transforming growth factor beta (TGFβ) and activator protein-1 [
6]. TGFβ is a multifunctional cytokine and improves wound healing by promoting fibroblast growth [
7,
8] and collagen I and III formation [
9] and by preventing matrix degradation through induction of protease inhibitors. There is a close correlation between the TGFβR-mitogen-activated protein kinases (MAPKs) [
10,
11]. The MAP kinases are grouped into three families, which are extracellular-signal-regulated kinases (ERKs), Jun amino-terminal kinases (JNKs), and stress-activated protein kinases (p38/SAPKs). Each cascade is initiated by specific extracellular and intracellular stimuli such as several stresses, growth factors, cytokines, hormones, oxidative stress and mitogens. It leads to activation of specific MAPK following activation of MAPK kinase kinase and MAPK kinase independently or dependently of each other. Activated MAPK phosphorylates several substrates in the cytosol and nucleus to occur about changes in protein function and gene expression that carry out the biological response including cell proliferation, differentiation, inflammation, and cell death in eukaryotes. And dysregulation of MAPK pathways has been implicated in many diseases.
Second, erythema is an abnormal skin condition characterized by redness following stresses, including exposure to heat, infections, allergens, and radiation (sunlight, ultraviolet, X-ray). Erythema is continued by late stage of wound healing after injury. Erythema is a rapid angiogenic response during wound healing in skin. Angiogenesis is a critical factor of the normal healing process. New blood vessels deliver oxygen, nutrients, and growth factors to injured tissues. On the other hand, erythema is a common side effect following treatment therapy such as laser procedures on skin [
12]. In these reason, postoperative cares, including cooling sprays, light-based modalities and dressings are conducted for a relief of erythema. Therefore, it is necessary to study an adjustable treatment for relief of erythema by modulation of thrombospondin 1 (TSP1) level, but dose not delay early stage of wound healing. TSP1 is major protein during angiogenesis and tumorigenesis [
13]. And it is a trimeric, calcium-binding molecule composed of several domains. Both positive and negative regulation of cell adhesion and growth have been influenced by to TSP1. TSP1 also have an effect on neuronal migration in the rostral migratory stream [
14]. Andrikopoulos et al. [
15] reported that voltage-gated ion channels may be novel targets for controlling angiogenesis [
16]. Wound healing might employ several possible mechanisms via ion channel modulation, including the inhibition of several processes: MAPK-dependent activation, the reception of anti-angiogenic TSP1, vascular endothelial growth factor (VEGF), pro-angiogenic factor and VEGF receptor expression, and matrix metalloproteinase inhibitor expression. However, the direct mechanism of wound healing by KCl-mediated ion channel function in skin remains unclear. Therefore, we showed that ion channel regulation, mediated by KCl, functions in wound healing as well as relief of erythema by regulation of TSP1 level.
METHODS
Cell culture
HS27 cells were grown to confluence in high-glucose dulbecco modified eagle medium with 10% fetal bovine serum supplemented with penicillin (100 units/mL) and streptomycin (100 mg/mL). Cells were cultured at 37℃ in a humidified atmosphere of 5% CO2.
Western blot
Cultured cells was rinsed with phosphate-buffered saline and incubated for 20 minutes in lysis buffer with 1 mM phenylmethanesulfonyl fluoride. After centrifugation at 14,000 rpm for 10 minutes, protein concentration was quantified by the Bradford protein assay. For immunoblotting, equal amounts of proteins were electrophoresed in 12% sodium dodecyl sulfate-polyacrylamide gel and transferred onto polyvinylidene difluoride membranes (Bio-Rad, Hercules, CA, USA) using a transfer apparatus (Bio-Rad). The membranes were blocked for 1 hour with 5% dry milk or bovine serum albumin before addition of the primary antibody, which was incubated overnight at 4℃. Chemiluminescence of immunoreactive bands was detected with the ECL Plus Kit (GE Healthcare, Buckinghamshire, UK). Densitometric quantification of bands was performed using Multi Gauge ver. 2.3 Software (Fuji Film, Tokyo, Japan). The antibodies for procollagen type-I, VEGF, basic fibroblast growth factor (bFGF), and β-actin were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). The MAPK Family Antibody Sampler Kit (measuring expression of the total- and phospho-MAPK proteins). The antibodies for TGFβ and TSP1 and anti-mouse, anti-rabbit, and anti-goat HRP-conjugated secondary antibodies were purchased from Abcam (Cambridge, MA, USA). The relative expression of each protein was quantified by densitometric measurement after western blotting with the Multi Gauge ver. 3.1 Software. All data were obtained from triplicate experiments.
Cell viability
To examine cell viability, HS27 cells were plated 24 hours before the experiment in 12-well plates at a density of 3×104 cells/well. The cells were then treated for 24 hours with KCl at several concentrations (0, 4, 8, 12, and 25 mM). Then, cells were incubated in growth medium with Cell Counting Kit-8 (CCK-8) Reagent (Dojindo, Tabaru, Japan). After incubation for 2 hours, absorbance of each sample was measured at 450 nm using spectrophotometer.
Chemicals
KCl was dissolved in water to prepare a stock 2.5 M solution for subsequent dilution. NS1619 (Sigma, St. Louis, MO, USA) was dissolved in dimethyl sulfoxide (DMSO) to prepare a stock 10 mM solution. Each chemical was added to the experimental solutions, and the final concentration of DMSO never exceeded 0.1%.
Statistics
All results are expressed as the means±standard error of at least three independent experiments. Statistical analyses were performed using Sigmaplot, and significant differences were assessed using the Student's t-test. When necessary, multiple comparisons were made using the Kruskal-Wallis test. (*P<0.05, **P<0.01, and ***P<0.001). NS indicates not significant.
DISCUSSION
Wound healing is a natural repair response to injury and involves complex interactions between cellular and molecular cascades. In this report, we focused on collagen formation and erythema related to ion channel-mediated signal pathways. Collagen is the major component of wound healing [
17,
18]. For this reason, natural or synthetic collagen in sponges, membranes, and dressings is used to anchor cytoactive agents for wound healing [
19]. During collagen synthesis in the proliferation phase of wound healing, various secreted cytokines and growth factors, such as TGFβ, bFGF, and platelet-derived growth factor, control the status of fibroblasts [
20,
21]. bFGF stimulates vascularization to increase the oxygen and nutrient supply that promotes collagen synthesis. The TGFβ signaling pathway is involved in many cellular processes, including cell growth and homeostasis, development and immune functions. TGFβ superfamily ligands phosphorylate receptor-regulated SMADs by binding to the extracellular fibroblast TGFβR [
22]. Alternatively, TGFβ activates non-Smad pathways, including the three MAPK pathways: ERK, JNK, and p38 MAPK [
22]. Ion channel activity modulates the MAPK pathways [
23]. Investigating wounded tissue repair is necessary to understand the biological and molecular determinants of tissue regeneration and to develop new methods to promote repair.
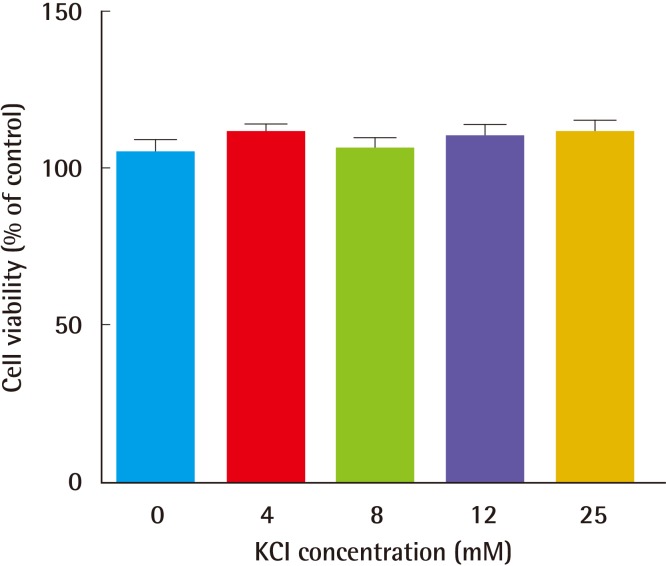
We hypothesized that KCl has wound healing properties, which are mediated by ion channel-induced MAPK cascades, and that these pathways regulate the balance between collagen synthesis and angiogenesis. First, we investigated whether alteration in KCl levels affected cell viability, and no change was observed in the viability of 25 mM KCl-treated HS27 cells (
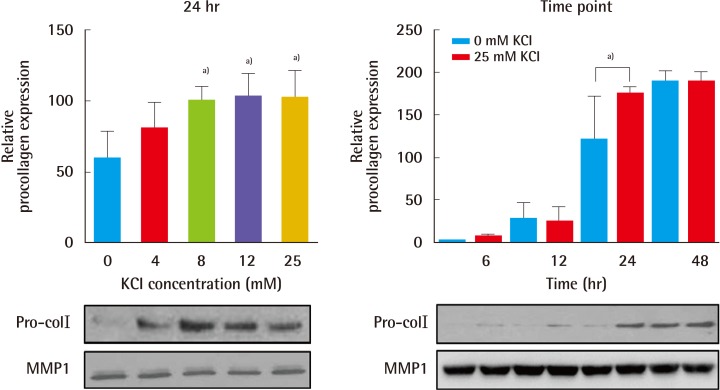
Fig. 1). Additionally, treatment with 25 mM KCl for 24 hours resulted in a 50% increase in procollagen levels (
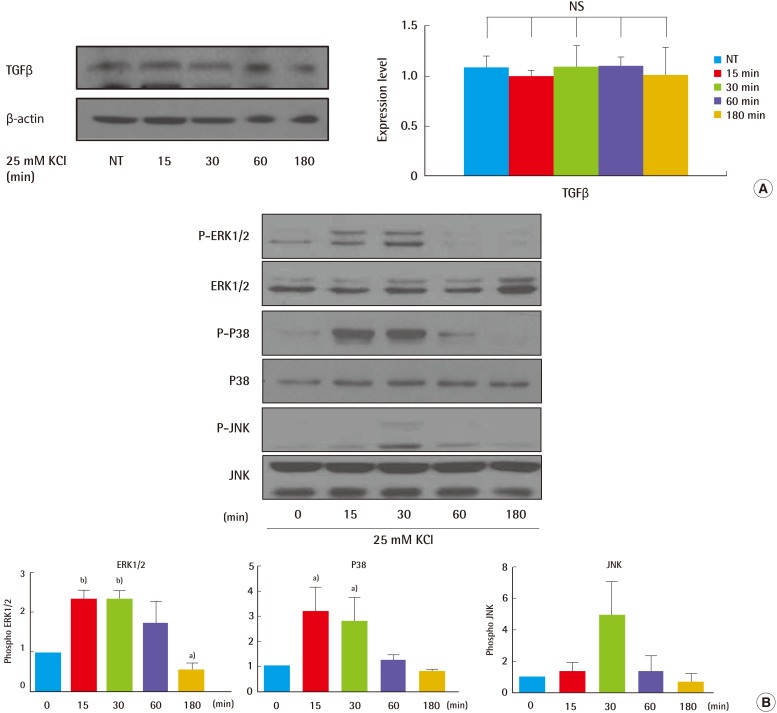
Fig. 2), showing that treatment with 25 mM KCl specifically contributed to collagen induction observed in human fibroblasts. As a result, we hypothesized that KCl may promote wound healing by increasing collagen synthesis without cell damage in the skin. To investigate the regulatory role of KCl in collagen synthesis, mediated by MAPK, we analyzed the expression of TGFβ signaling-related proteins, such as ERK, p38, JNK, in untreated and KCl-treated cells, and we assessed how KCl treatment altered levels of signaling proteins upstream of collagen synthesis (
Fig. 3). K
+ channels may control epithelial repair processes [
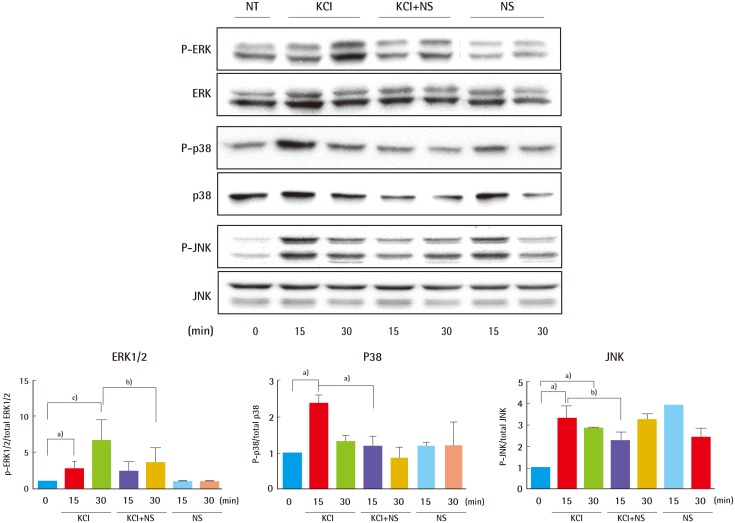
24]. Compared with cells treated with KCl alone, cells treated with KCl after NS1619 pre-treatment for 30 minutes showed 45% and 50% decreases in phosphorylated ERK1/2 and p38 (
Fig. 4). Thus, KCl may promote collagen synthesis by modulation of ion channel-specific activation of MAPK pathways, independently of the TGFβR.
Angiogenesis, new blood vessel formation, is necessary for wound healing but too much angiogenesis result in erythema in impaired skin. We hypothesized that KCl may function in wound healing by regulating the balance of collagen formation and angiogenesis. TSP1 was firstly identified endogenous inhibitor of angiogenesis, and it is a multifunctional, extracellular matrix protein [
15]. Further, TSP1 is critical in wound healing. TSP1 expression is typically low in normal skin, is induced acutely after wounding, and is diminished by about 14 days post-wounding [
16]. Recently, it was also reported that the transient potential receptor channel 4 controls TSP1 secretion and angiogenesis in renal cell carcinoma [
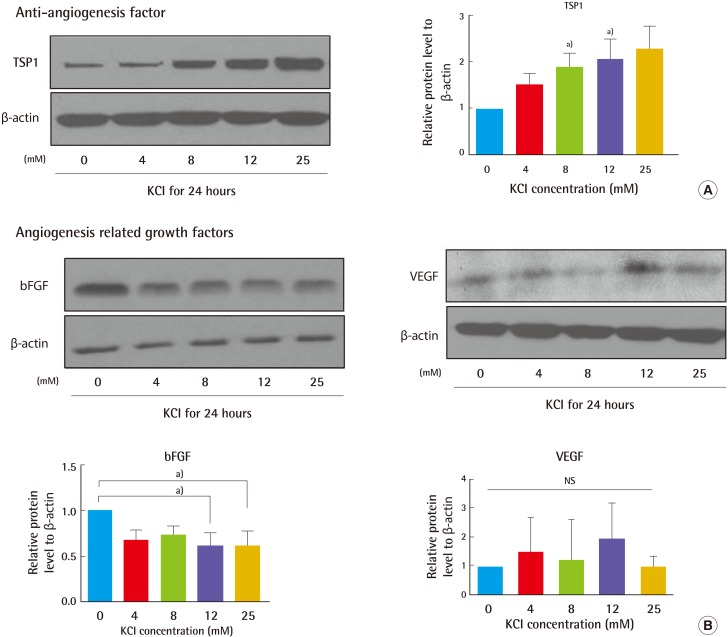
25]. Therefore, we investigated whether KCl regulates TSP1 expression, mediated by wound healing signaling pathways. KCl-treated cells had increased TSP1 levels, dependent on KCl concentration. bFGF protein levels were decreased in the KCl-treated cells (
Fig. 5). In KCl-treated cells, the induction of TSP1 might be a negative feedback mechanism to counterbalance the effect of pro-angiogenic factors such as bFGF and VEGF.
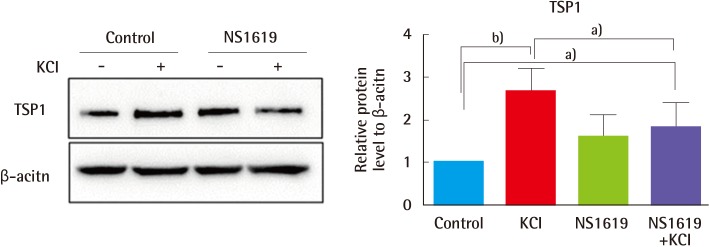
Finally, we considered the possibility that regulation of ion channels by KCl causes upregulation of TSP1 protein, which is involved in angiogenesis and wound healing. KCl-treated cells exhibited induction of TSP1 levels, which would lead to anti-angiogenic effects. The TSP1 induction was reversed by NS-1619, a specific K
+ channel opener (
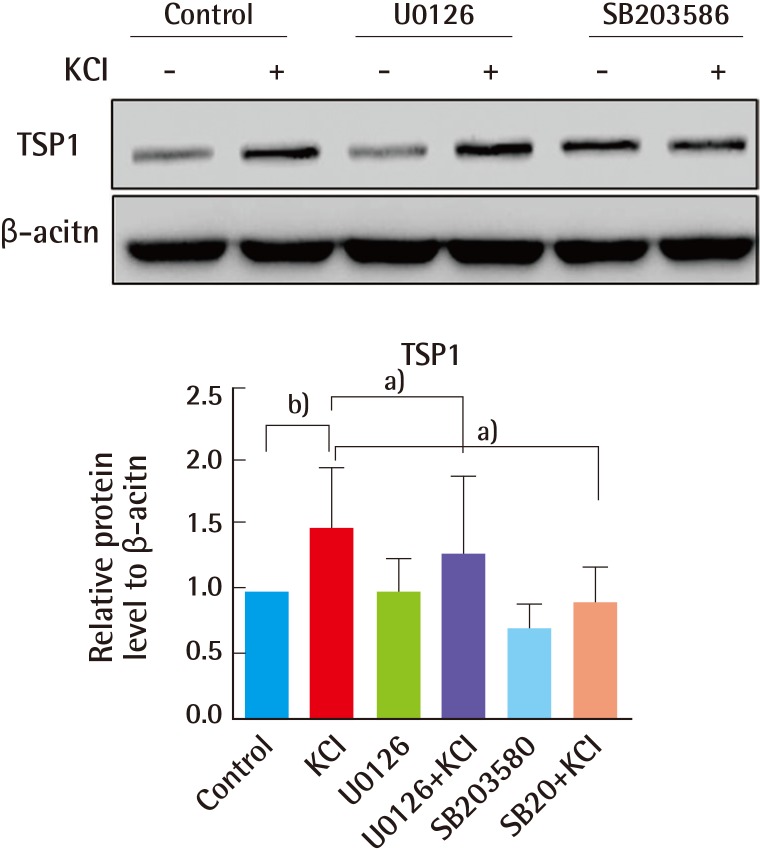
Fig. 6). To assess the role of this signaling pathway in the regulation of TSP1 by KCl, cells were treated with inhibitors against ERK (U0126) and p38 (SB203585). TSP1 levels were increased in KCl-treated cells, and increased TSP1 expression was downregulated after pre-treatment with ERK and p38 inhibitors (
Fig. 7). Collectively, we suggest that KCl treatment may relieve angiogenesis, by inducing TSP1 expression, through ion channel-mediated ERK and p38 MAPK signaling, not TGFβ signaling.
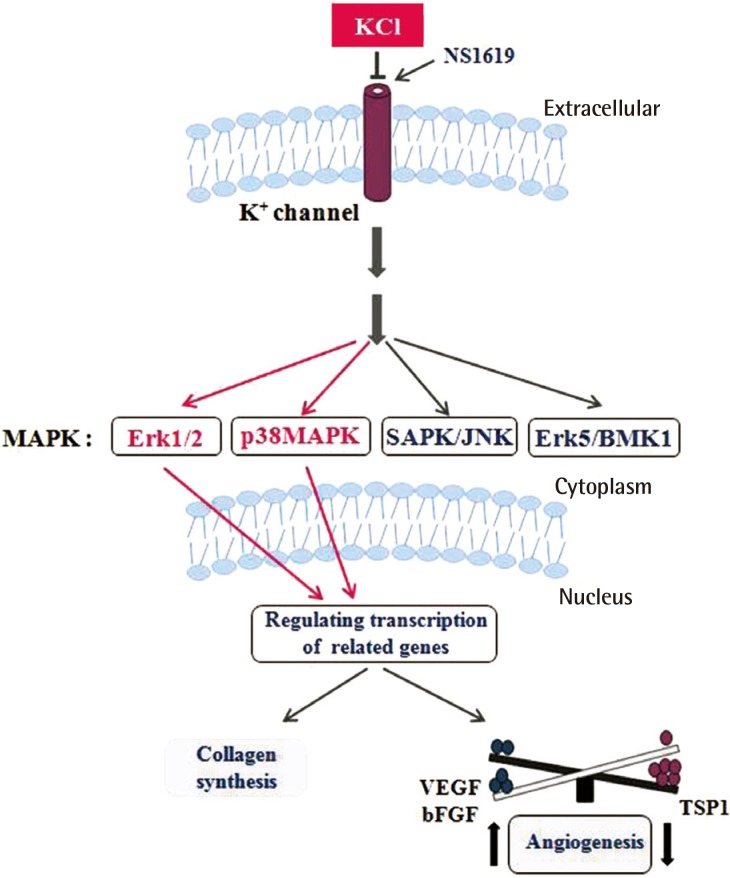
Collagen and TSP1 expression increased significantly in KCl-treated cells than in untreated cells. Collagen which is produced by KCl helps to skin's regeneration. Erythema that result by angiogenesis after wound decreases by treating KCl with induction of TSP1 level. During wound healing, the skin often shows erythema, which is caused by angiogenesis; nevertheless, angiogenesis supplies the essential elements for the remaining healing process. In addition, we established the mechanism of collagen production and inhibition of angiogenesis through the ERK and p38 pathways, which are mediated by regulation of K
+ ion channels (
Fig. 8), Taken together, these findings indicate that the appropriate concentration of KCl modulates the balance between anti- and pro-angiogenic mediators, leading to collagen synthesis and erythema maintaining moderate level of angiogenesis during wound healing process (
Fig. 8). The results of the present study will provide important insight on the wound healing mechanism of KCl via ion channel-mediated MAPK signaling in animal model with adjust concentration of KCl. In addition, this finding may lead to the ideal application of collagen synthesis-based therapies to help modulate balance of angiogenesis in human skin.