DISCUSSION
Inflammation, proliferation, and maturation are distinct phases in the progression of wound healing, displaying a regression curve over time with a graded effect [
15]. Wounds are further categorized as acute, chronic, or chronic nonhealing, the last of these marked by >6 months of conventional routine wound care. Conditions favoring delayed wound healing include advanced age, diabetes mellitus (DM), sustained pressure, vascular insufficiency, renal failure, jaundice, infection, and immunosuppression. In older patients, a multiplicity of these conditions is apt to be at play. Conservative treatment may be the only recourse for patients taking prohibitive medication (such as aspirin) or whose overall status is unacceptable for surgical management.
In the case of large wounds, infections are difficult to control, and therefore, the wounds may themselves contribute substantially to the general deterioration of the already weakened patients. Although conservative treatment with antibiotics and dressings may control infections, the emergence of multidrug-resistant bacterial strains has limited this approach. In some circumstances, patients may only be candidates for dressing changes in isolation, with no chances of debridement and reconstructive surgery. This further impacts in-hospital infection rates and durations of hospital stay, adding to the burden of healthcare.
Normal skin pH (range, 4-6) is weakly acidic, due to the amino and lactic acid contents of the horny layer and cutaneous adnexa. The acidity of skin contributes to its barrier function [
16]. In chronic open wounds, a weakly alkaline pH range of 7.15-8.9 (8.0 on average) encourages the growth of ammonia-producing and CO
2-generating bacteria [
16,
17,
18]. Ammonia is directly toxic to wounds, inflicting injury through heightened tissue protease activity. Tissue ischemia then ensues, marked by elevated saturated oxyhemoglobin and diminished oxygen partial pressure. Greater acidity helps to reverse these effects [
18]. Ultimately, an acid environment is destructive to bacteria and normalizes the wound milieu. NH
3- is reduced to NH4, thus restoring normal protease activity and encouraging healing [
17].
The importance of skin pH was first underscored by the addition of acetic acid to a honey-based remedy (Medihoney, Medihoney Ltd., Queensland, Austria) approved in Europe for the treatment of chronic wounds (venous ulcers, DM foot, and burns) [
2]. Dressings soaked in 5% dilute acetic acid and applied to wounds with pseudomonas infections (
P. aeruginosa) produced positive clinical results in case studies during the 1960s [
6]. However, the short product half-life (<1 hr), necessitating frequent dressing changes, was a clear disadvantage. Ryssel et al. [
7,
8,
9] studied the
in vitro sensitivity of pseudomonas strains commonly encountered in burn centers, confirming the strong antimicrobial effects of acetic acid on
P. vulgaris,
P. aeroginosa, and
A. baumanii-organisms often present in persistent wounds, despite long-term antibiotic therapy [
7,
8,
9]. MRSA proved sensitive as well.
The high viscosity of honey is also beneficial, eliminating any exudates present and otherwise creating a damp environment optimal for wound healing. Keratinocyte and fibrocyte survival rates are enhanced under such conditions. Honey also contains glucose oxidase, which adds acidity through the conversion of glucose to gluconic acid (measured pH=3.5). Acidity alters biofilms that block the delivery of drugs to chronic wounds and exerts antibacterial effects. Aided by bacterial polysaccharide and exudates, biofilms adhere to wound surfaces, are difficult to remove, and serve as reservoirs for continuous microbial release [
2]. According to recent reports, a majority (65%) of infectious diseases are attributable to biofilms [
19].
It is clear that acetic acid promotes wound healing, enzymatic activity, and immunity, and attenuates microbial proliferation and biofilm virulence [
18]. However, olfactory stimulation (at least initially) and the need for frequent dressing changes are notable drawbacks. At a higher acidic concentration (for greater antibiotic effect), patients will experience more pain [
7].
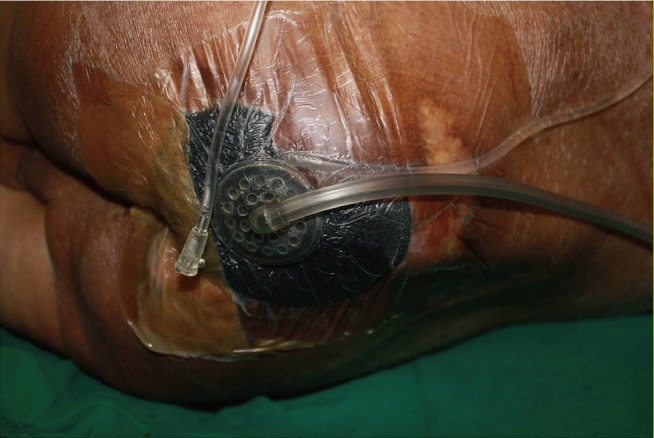
Nonoperative NPWT of the existing complex defects is now widely used, owing to national medical insurance coverage (2010) and miniaturization of equipment. The vacuum-assisted closure device (Kinetics Concepts Inc.), consisting of a porous polyurethane sponge (applied to wounds) for moisture and a vacuum-connected film dressing, was first introduced in 1997 under negative pressure, exudates are extracted, the blood supply is increased, and fewer dressing changes are required [
3,
5]. As explained by Scherer et al. [
5], negative pressure boosts collagen synthesis and neovascularization by wound transformation (macrodeformation) and three-dimensional migration (microdeformation), both encouraging DNA synthesis [
5]. In 2011, de Laat et al. [
3] found that wound healing was two times faster with NPWT (vs. sodium hypochlorite) in a prospective randomized study, and Kim et al. [
4] reported a 95% rate of limb salvage with NPWT of the DM foot.
In according to international guide line on NPWT with instillation, debridement of biofilms and necrotic tissues is essentially needed and provide to favorable pH milieu for wound healing [
20,
21]. In addition, once weekly debridement tends to restart the wound-healing cycle, interrupting fibroblast senescence at the inflammatory stage [
15]. However, patients who have DM feet, pressure sores, and venous ulcers and who are taking anticoagulants or lack ready access to facilities may forego this step. Consequently, the outcomes of NPWT may fall short of expectations. Furthermore, most of these patients harbor serious hospital-acquired infectious organisms, such as MRSA, vancomycin-resistant
Enterococcus, and carbapenem-resistant gram-negative bacilli (
A. baumani,
P. areuginosa, and
K. pneumoniae). In 2009, Arias and Murray [
1] reported on multi-drug resistant strains and the use of antibiotics in the 21st century, emphasizing that antibiotic choices for these strains are few and carry toxicities.
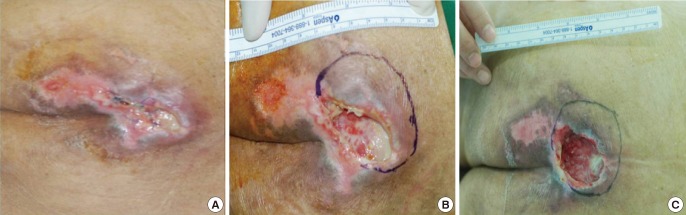
Our approach combines the convenience of NPWT with the infection control of acetic acid irrigation. The three patients that we treated had infected wounds, with excessive exudation, local redness, warmth, poor granulation tissue, and foul odor. Given the sacral locations, fecal contamination was likely and proved difficult to manage. The wound sites failed to improve, with infectious symptoms persisting, despite three weeks of conventional wound care. Although film dressings are essential to avoid fecal contamination, the increased CO2 of a closed system is problematic, which is why vacuum dressings are vulnerable to infection. Infection control is mandatory for wounds of this nature in order to ensure that healing (i.e., granulation tissue and surface vascularization) takes place.
NPWT and instillation have been combined in many earlier attempts, but guidelines for the same have been set recently [
20]. We used twice daily 1% acetic acid irrigation, delivered in 30-mL aliquots over a period of 15-30 minutes and it differs little from the guideline. A reduction in wound pH resulted (strongly alkaline at baseline; weakly alkaline or neutral post-treatment) [
22,
23]. In addition, all pathogenic bacteria identified in wound cultures (MRSA,
P. mirabilis,
C. albicans) dissipated or cleared. Unlike antibiotic therapy, this approach effectively controlled infection, irrespective of the culpable organisms.
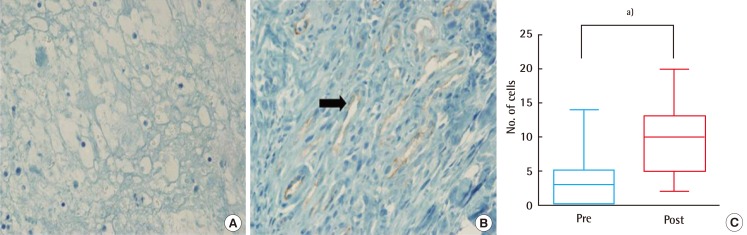
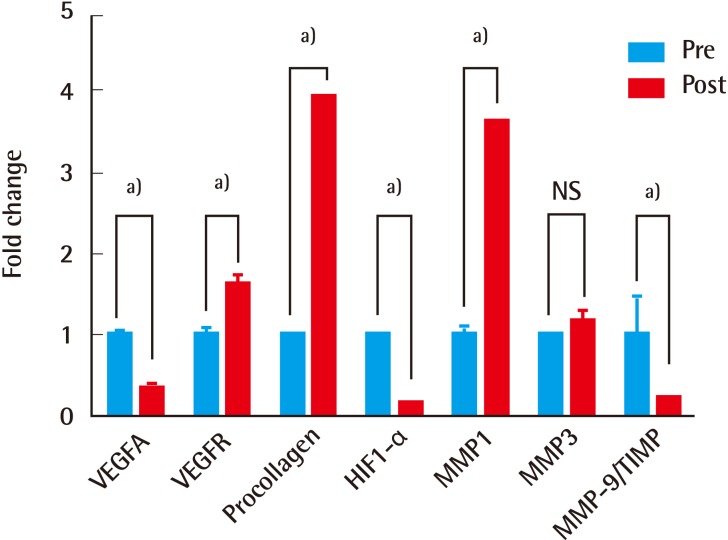
In chronic wounds, MMP-9/TIMP ratios appear to reflect the actual proteolytic environments, more so than either determinant does alone. The significant decline in the MMP-9/TIMP ratio (based on the RT-PCR assay) seen in our patients post-treatment suggests abatement of prolonged inflammation and wound chronicity [
14,
24,
25]. Indeed, the odor grew less intense on physical exams and the exudates retracted visibly, indicating that infection control is clearly fundamental to wound healing.
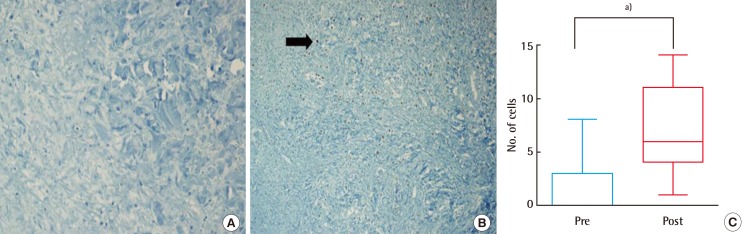
Re-epithelialization (from the edges of soft tissue defects) and angiogenesis are enabled by the migration of fibroblasts along fibrin networks. Factors promoting inflammation (IL-1a, IL-1b, IL-6, and tumor necrosis factor a), facilitating collagen synthesis (fibroblast growth factor [FGF]-2, insulin-like growth factor-1, and transferring growth factor b [TGF-b]), converting fibroblasts to myofibroblasts (TGF-b), and initiating angiogenesis (EGF, FGF-2, and TGF-a), are released by platelets and white blood cells at the wound sites. The latter also aid in re-epithelialization. In our patients, significant post-treatment increases in CD31 staining and VEGFR expression (vs. baseline levels) corresponded with enhanced tissue vascularization. Ki-67 staining and procollagen levels also increased significantly, reflecting the active proliferation of fibroblasts and collagen synthesis. Proliferation and migration of keratinocytes at wound sites (for re-epithelialization) were similarly boosted, as indicated by a significant increase in MMP-1 after therapy. Finally, HIF-1-alpha (a gauge of tissue hypoxia) significantly declined following treatment.
At wound margins, cells must loosen cell-to-cell and cell-to-extracellular matrix contacts in order to migrate and re-epithelialize wound surfaces. Multiple MMPs have been implicated from this aspect of wound repair, including MMP-1, -3, -7, -9, -10, -14, and -28. MMP-1 (collagenase-1) is present in human skin wounds during re-epithelialization but diminishes once closure is complete. The expression of MMP-1 is stimulated by the binding of integrin to type-I collagen (abundant in the dermis). MMP-3 (stromelysin-1) is concentrated in cells behind the migrating front [
11]. In our test patients, a statistically significant post-treatment increase in MMP-1 (vs. baseline) signaled that the process of re-epithelialization was well underway [
11]. Moreover, reductions in surface areas (average, 35%) and capsule sizes (average, 29%) of wounds were documented.
Our experiment used highly infected wounds (high risk) to which NPWT with just black polyurethane ether foam, Granu-Foam (Kinetics Concepts Inc., San Antonio, Texas, USA) cannot be applied in the first place. Further, we believe that a combination of NPWT and acetic acid irrigation can change the infected wound from high risk to low risk and help enhance collagen synthesis, angiogenesis, and inflammation control, and have proved it from the perspective of molecular biology. Moreover, we gathered tissue specimens that were used in immunohistochemistry and quantitative polymerase chain reaction instead of the collected fluid.
From our perspective, combining a vacuum dressing with acetic acid irrigation is beneficial in many ways. Although film dressings help prevent fecal contamination (particularly in pressure sores), extensive wound irrigation and debridement should be carried out anyway with each dressing change. Acidic wound irrigation unquestionably reduces the bacterial burden, and when combined with NPWT, the protease-induced inflammatory phase of wounds is stabilized, lowering the MMP-9/TIMP ratio. Similarly, the acidification of alkaline wounds reduces ammonia toxicity, creating a more favorable wound milieu (i.e., reduced saturation of oxyhemoglobin and increased partial pressure of oxygen).
Used alone, NPWT may increase angiogenesis, but adding 1% acetic acid soaks enhances re-epithelialization via the upregulation of MMP-1. Through macro- and microdeformation, NPWT also continuously encourages the proliferation of fibroblasts, albeit fibroblast senescence is averted by debridement too [
15]. Because chronically infected soft tissue defects are not always amenable to immediate reconstructive surgery, our protocol provides another means to reduce wound size and control infection, allowing time for a patient's general condition to improve.
Admittedly, the findings of this study are preliminary and are based on the few patients studied. However, further research is warranted in this setting, recruiting more patients of this type for similar investigations. NPWT and acetic acid irrigation, when used jointly, help decrease the pH of chronic wounds, reducing their size and resolving infection so that wound healing may proceed.